Study Notes

Overview
Welcome to your deep dive into Group 1 of the periodic table, the Alkali Metals. This topic is a cornerstone of inorganic chemistry at GCSE and is frequently tested by examiners. It explores the relationship between electronic structure and physical and chemical properties, a fundamental concept that underpins much of chemistry. You will learn why these metals are so reactive, how their reactivity changes as you go down the group, and what happens when they react with water and oxygen. A solid understanding here will not only secure marks on this topic but also provide a strong foundation for understanding other groups in the periodic table, particularly the Halogens in Group 7, as they are often compared. Typical exam questions range from short-answer recall of properties to longer, 6-mark questions requiring you to explain the trend in reactivity in detail.
Key Concepts
Concept 1: Atomic Structure and Properties
The defining feature of the Alkali Metals (Lithium, Sodium, Potassium, etc.) is their atomic structure. Every atom in this group has one electron in its outermost shell. This single outer electron is what dictates their chemical properties. To achieve a stable, full outer shell, they readily lose this electron, forming a positive ion with a +1 charge (e.g., Na⁺). This eagerness to lose an electron makes them very reactive. Physically, they are not typical metals; they have low densities (Li, Na, and K float on water) and are very soft, easily cut with a knife. They also have relatively low melting and boiling points that decrease as you descend the group.
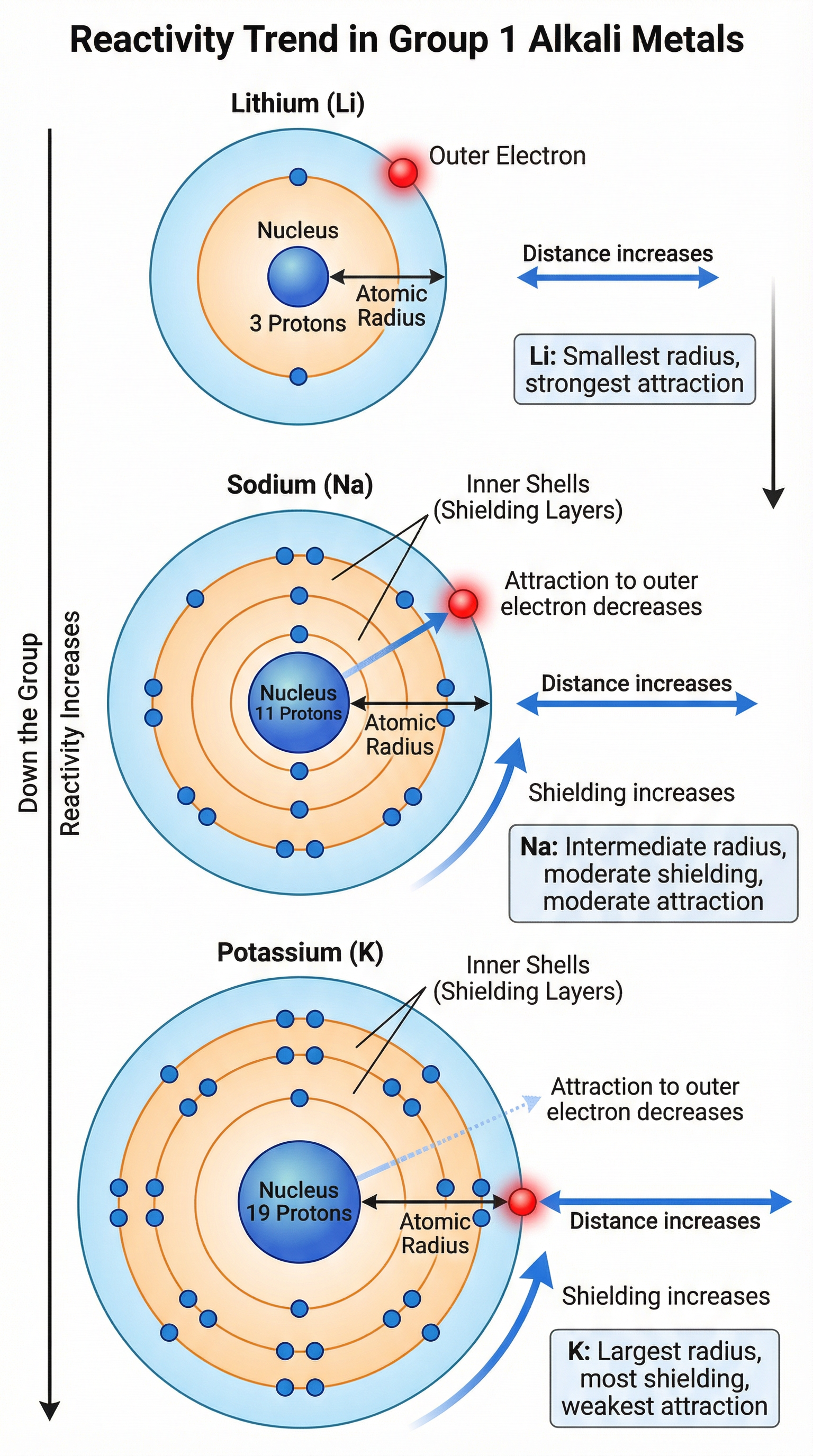
Concept 2: The Trend in Reactivity
This is the most important concept to master. Reactivity increases as you go down Group 1. A 6-mark question asking you to explain this is very common. Your answer must be structured around three key points:
- Atomic Radius Increases: As you go down the group, each element has an additional shell of electrons. This means the atom gets bigger, and the outermost electron is further from the nucleus.
- Shielding Increases: The inner shells of electrons shield the outer electron from the positive charge of the nucleus. The more shells there are, the greater the shielding effect.
- Nuclear Attraction Decreases: The combination of the greater distance and increased shielding means that the electrostatic force of attraction between the positive nucleus and the single outer electron gets weaker. Because this attraction is weaker, less energy is needed to remove the outer electron. Therefore, the atom can lose its outer electron more easily, making it more reactive.
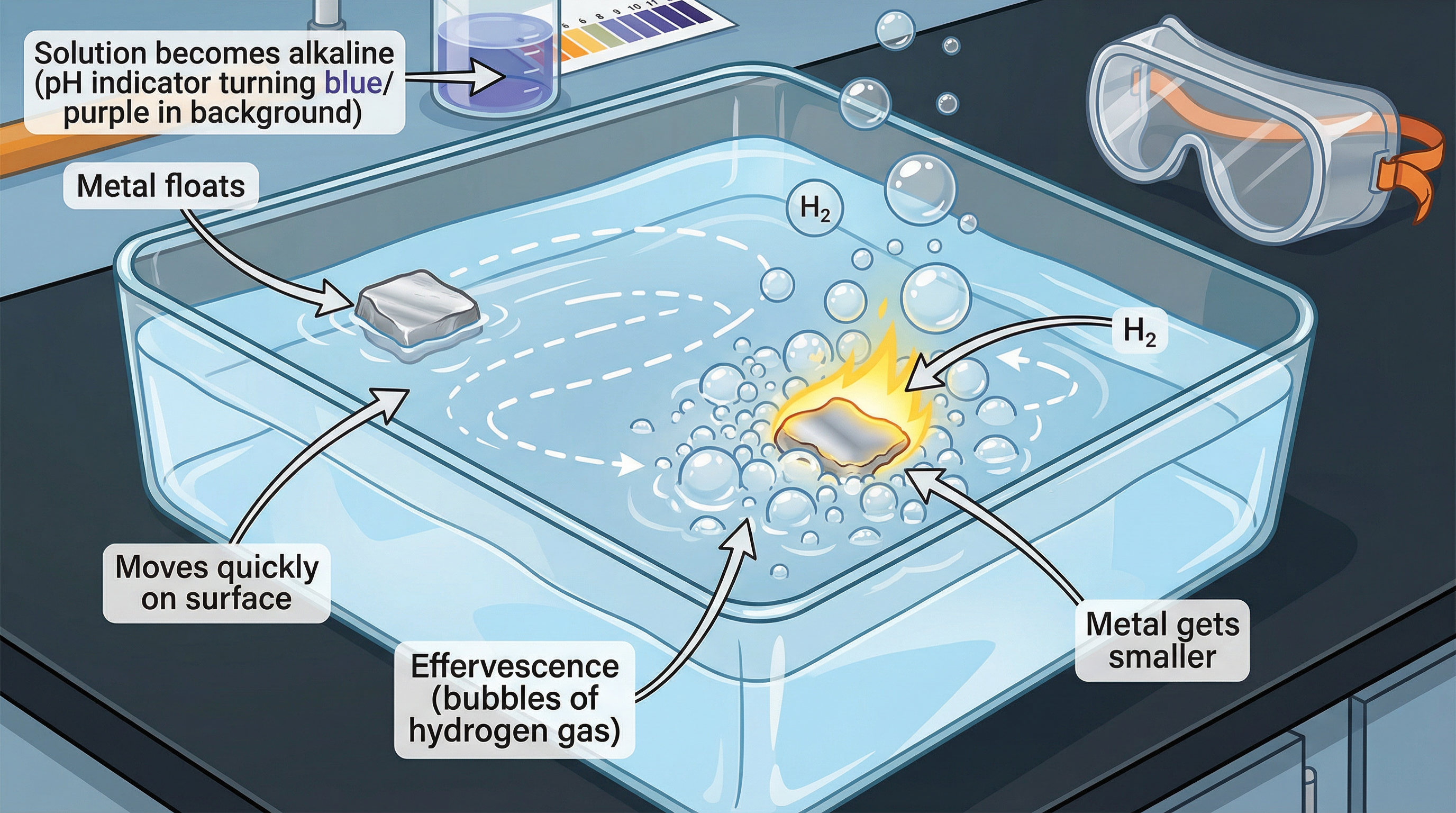
Concept 3: Reactions with Water
The reaction of alkali metals with water provides clear evidence for the trend in reactivity. The general equation for the reaction is:
2M(s) + 2H₂O(l) → 2MOH(aq) + H₂(g)
Where 'M' represents any alkali metal.
Two key products are formed: a metal hydroxide (which is an alkali, hence the group name) and hydrogen gas. The observations become more vigorous down the group:
- Lithium: Floats, fizzes steadily, moves around the surface.
- Sodium: Fizzes rapidly, melts into a silvery ball, darts across the surface.
- Potassium: Reacts violently, the hydrogen gas produced ignites instantly and burns with a lilac flame.
Listen to our Podcast!
Mathematical/Scientific Relationships
Balanced Symbol Equations
Being able to write balanced symbol equations is crucial. You must include state symbols to gain full marks.
- Reaction with Water:
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g) - Reaction with Oxygen:
4Na(s) + O₂(g) → 2Na₂O(s) - Reaction with Chlorine (a Halogen):
2Na(s) + Cl₂(g) → 2NaCl(s)
Practical Applications
Flame Tests
A key required practical is using flame tests to identify metal ions. When alkali metal compounds are heated in a flame, they produce characteristic colours. This happens because the heat gives energy to the electrons, causing them to jump to higher energy levels. When they fall back down, they release energy as light of a specific colour.
- Apparatus: Bunsen burner, nichrome wire loop, hydrochloric acid (for cleaning the loop), samples of metal compounds.
- Method: Dip the clean wire loop into the sample and place it in a blue Bunsen flame. Observe the colour.
- Results:
- Lithium (Li⁺): Red
- Sodium (Na⁺): Yellow/Orange
- Potassium (K⁺): Lilac (Pink)
- Examiner Tip: The lilac flame of potassium can be difficult to see, especially if contaminated with sodium (which is very common). Examiners accept that you can observe the flame through a blue cobalt glass, which absorbs the yellow light from sodium, making the lilac flame visible."