Study Notes

Overview
Ethics are the moral principles and rules of conduct that guide psychologists when they conduct research. For the OCR GCSE Psychology exam, candidates are expected to have a firm grasp of the British Psychological Society (BPS) guidelines and be able to apply them to novel research scenarios. This is not just about memorising definitions; it is about understanding the "why" behind each principle and the practical steps researchers must take to protect their participants. Historically, studies like Milgram's obedience experiments highlighted the potential for psychological distress, leading to the formalisation of these essential rules. Examiners will credit responses that move beyond simple identification of issues to a nuanced evaluation of research design and the proposal of specific, workable solutions. This guide will equip you with the knowledge and exam technique to confidently tackle any ethics-related question.
The Core BPS Ethical Guidelines

1. Informed Consent
What it is: Before a study begins, participants must be given comprehensive information about the aims, procedures, potential risks, and their right to withdraw. Based on this information, they must freely agree to participate. For participants under 16, parental consent is required.
Why it matters: This principle respects individual autonomy. It ensures that people are not coerced or tricked into participating in research they would otherwise refuse. In the exam, credit is given for explaining that consent must be 'informed', not just assumed.
2. Deception
What it is: Deliberately misleading or withholding information from participants about the true nature of the study. The BPS states deception should be avoided unless it is strongly justified by the study's scientific value and there is no alternative.
Why it matters: Deception can lead to distrust in psychology and may cause participants distress upon discovering the true aim. A common mistake is to state that deception is banned. It is not, but it must be followed by a full debriefing.
3. Protection from Harm
What it is: Researchers have a responsibility to protect participants from any physical or psychological harm that is greater than what they might experience in their everyday lives. This includes stress, embarrassment, anxiety, or damage to self-esteem.
Why it matters: This is the cornerstone of ethical research. Candidates must be able to distinguish between physical harm (e.g., injury) and psychological harm (e.g., distress from an unsolvable puzzle) and apply this to the stimulus material.
4. Right to Withdraw
What it is: Participants must be informed at the start of a study that they can leave at any point, without giving a reason and without facing any penalty. They can also request that their data be destroyed after the study has concluded.
Why it matters: This empowers the participant and acts as a safeguard. If a participant feels uncomfortable, they can exit the situation. In Milgram's study, the experimenter's prods like "You have no choice, you must go on" were a clear violation of this right.
5. Confidentiality
What it is: The communication of personal information from one person to another with the trust that it will not be revealed to others. In research, this means participant data is kept private and secure.
Why it matters: People are more likely to be honest in their responses if they know their information will be protected. This is often confused with anonymity. Confidentiality means the researcher knows the identity but protects the data; anonymity means the researcher does not know the identity.
6. Privacy
What it is: A person's right to control the flow of information about themselves. In research, this means not observing people in situations where they would not expect to be observed (e.g., in their home) without their consent.
Why it matters: This respects personal boundaries. Public observation (e.g., in a shopping centre) is generally considered acceptable, but the context is always key. Examiners will reward answers that consider the specific setting described in the stimulus.
Dealing with Ethical Issues
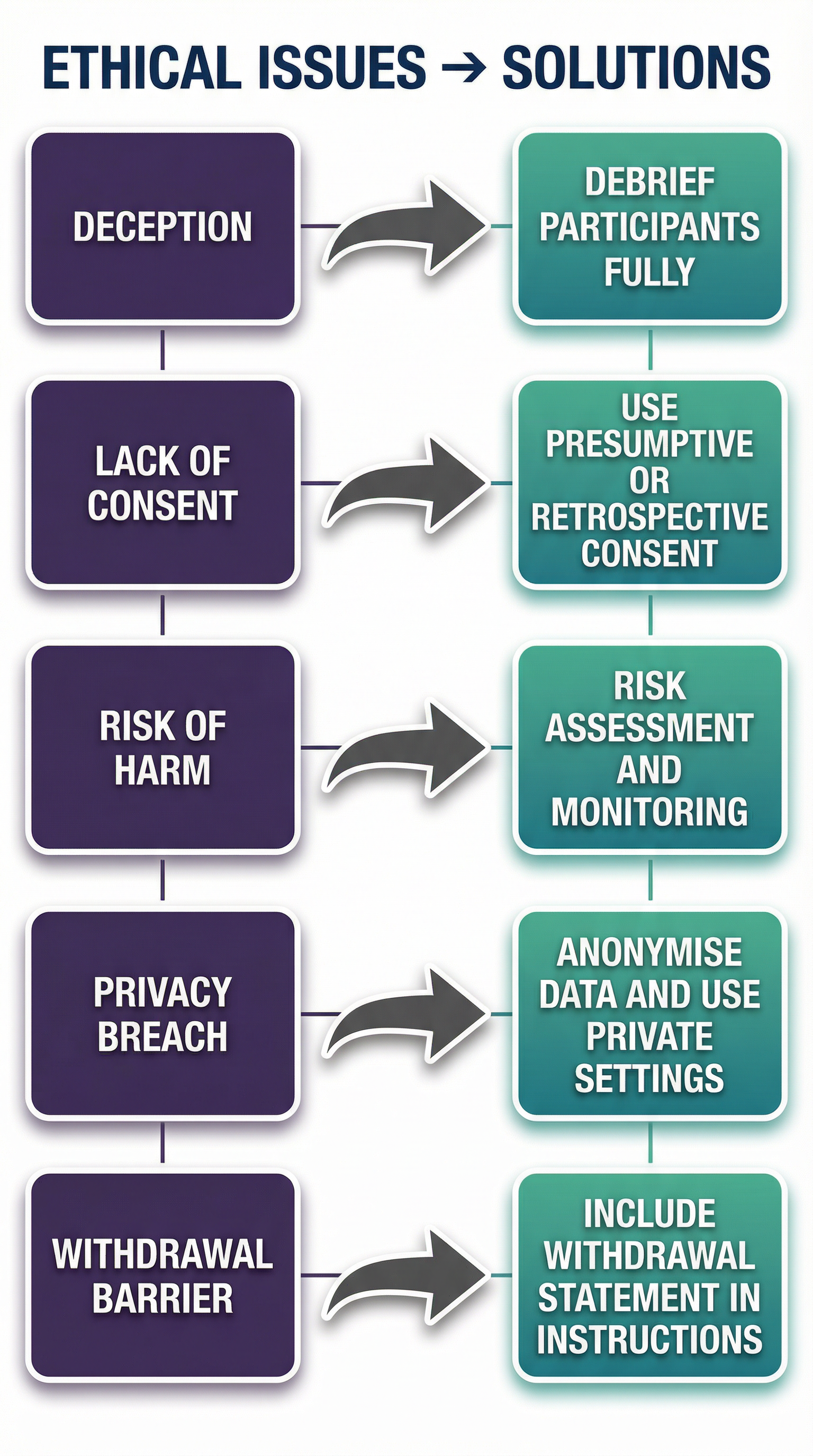
Examiners want to see practical solutions. For every ethical issue you identify, you must be able to suggest a way to 'deal with' it.
- For Deception: Conduct a full Debrief. This involves revealing the true aim, explaining why deception was necessary, and providing an opportunity for questions. The goal is to return the participant to the state they were in before the study.
- For Lack of Informed Consent: Use Presumptive Consent. This involves asking a similar group of people if they would find the study acceptable. If they agree, the researcher can 'presume' the actual participants would too. Alternatively, Retrospective Consent can be sought after the study is complete.
- For Risk of Harm: Conduct a thorough Risk Assessment. The researcher must anticipate potential risks and have measures in place to minimise them. This could involve screening participants or having a clinical psychologist on hand.
- For Confidentiality: Anonymise the data. This means removing any identifying information, such as names or addresses, and often involves assigning participants a number or code instead.