Study Notes
Overview
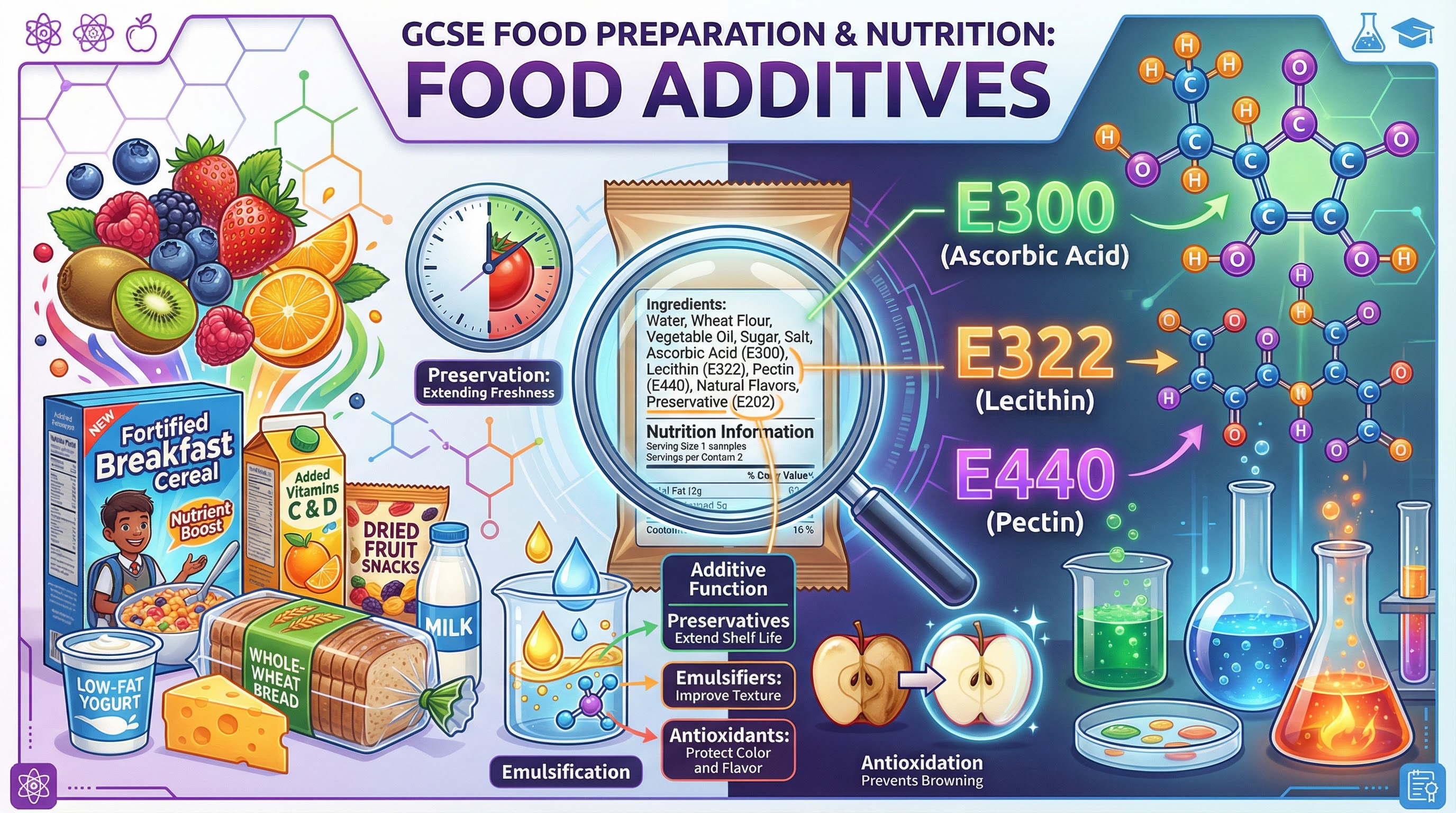
Food additives are substances added to products to perform specific technological functions, such as preserving freshness, improving texture, or enhancing appearance. For the OCR J309 specification, candidates must move beyond a simple definition and demonstrate a robust scientific understanding of how these additives work. Examiners expect you to analyse the technological necessity of additives against potential consumer health concerns, using precise terminology and specific, named examples. This topic requires you to link the chemical properties of an additive to its function, for instance, explaining how the molecular structure of an emulsifier allows it to bind with both oil and water. High-level responses will consistently evaluate the trade-offs involved in food production, such as extending shelf-life versus the risk of allergic reactions. This guide will equip you with the detailed knowledge and analytical skills required to excel.
Key Additive Groups
To gain credit, candidates must be able to categorise, explain, and give examples for the main additive groups. Vague answers stating additives 'improve food' will not be awarded marks.
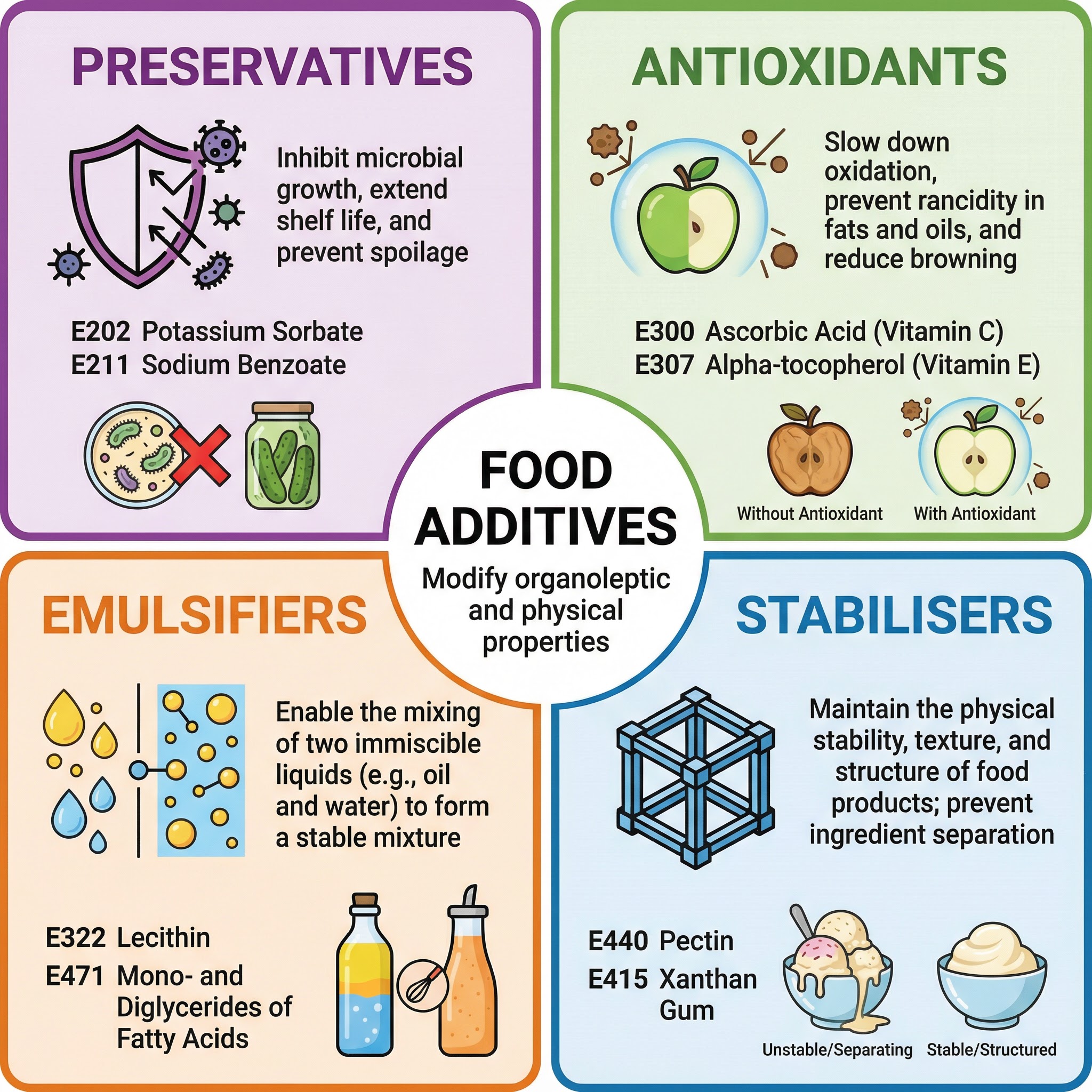
1. Preservatives
Function: To inhibit the growth of pathogenic microorganisms like bacteria, yeasts, and moulds, thereby extending the shelf-life of a product and preventing foodborne illness. They are a critical defence against food spoilage.
Mechanism: Preservatives work by creating an environment that is hostile to microbes. This can be by altering the pH of the food or by interfering with the microbe's cell membrane or enzymes.
Specific Knowledge:
- Natural Example: Salt (Sodium Chloride) used in curing meats.
- Artificial Example: Sodium Benzoate (E211), commonly used in acidic foods like carbonated drinks and jams.
2. Antioxidants
Function: To prevent or slow down the process of oxidation. Oxidation causes fats and oils to become rancid (oxidative rancidity) and leads to the enzymatic browning of cut fruits and vegetables.
Mechanism: Antioxidants are reducing agents that readily donate electrons, neutralising free radicals before they can damage the food molecules. This protects the food's flavour, colour, and nutrient content.
Specific Knowledge:
- Natural Example: Ascorbic Acid (Vitamin C, E300), found in citrus fruits.
- Artificial Example: Butylated Hydroxytoluene (BHT, E321), often used in breakfast cereals and fat-based products.
3. Emulsifiers
Function: To enable the mixing of two or more immiscible liquids, such as oil and water, to form a stable mixture known as an emulsion.
Mechanism: Emulsifier molecules are characterised by having a hydrophilic ('water-loving') head and a hydrophobic or lipophilic ('water-hating' or 'oil-loving') tail. The hydrophilic head is attracted to the water molecules, while the hydrophobic tail is attracted to the oil droplets, forming a stable bridge between them and preventing them from separating.
Specific Knowledge:
- Natural Example: Lecithin (E322), found in egg yolk and soybeans. It is essential for making mayonnaise.
4. Stabilisers
Function: To help maintain the uniform dispersion of substances in a food. They provide a smooth, consistent texture and prevent ingredients from separating out.
Mechanism: Stabilisers are often large molecules that form a network or gel structure within the food, trapping other molecules and preventing them from moving around. This gives the product 'body' and a stable structure.
Specific Knowledge:
- Natural Example: Pectin (E440), a polysaccharide extracted from citrus peel and apples, used to set jam.
- Artificial Example: Xanthan Gum (E415), used to thicken sauces and in gluten-free baking.
Natural vs. Artificial Additives
A common misconception is that all food additives are synthetic 'chemicals'. Examiners award credit for candidates who can distinguish between natural and artificial sources and understand that both are subject to the same rigorous safety testing by the Food Standards Agency (FSA).
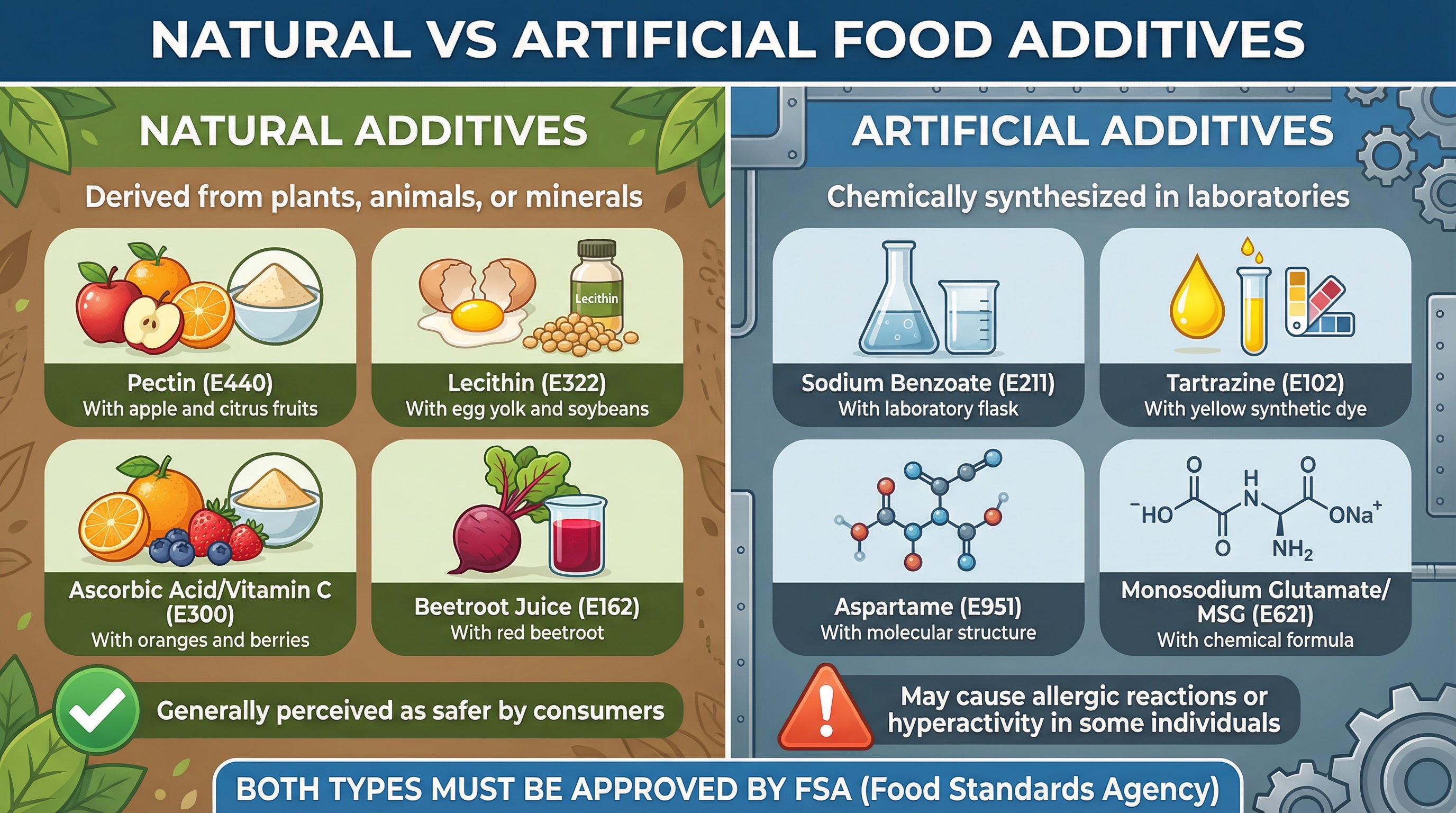
- Natural Additives: Derived from natural sources like plants (e.g., Beetroot Juice, E162, for colour), animals, or minerals. Consumers often perceive them as safer, but they can still cause allergic reactions in some people.
- Artificial Additives: These are synthesised chemically in a laboratory to mimic natural substances or to create compounds not found in nature. They are often cheaper to produce and more stable. However, some, like certain artificial colourings (e.g., Tartrazine, E102), have been linked to hyperactivity in children.