Study Notes

Overview
This study guide provides a comprehensive overview of the scientific principles underpinning sauce making, as required by the OCR J309 specification. Candidates are expected to move beyond simple recipe recall and demonstrate a robust understanding of the functional properties of ingredients. This includes the process of gelatinisation in starch-based sauces, the role of emulsifiers in creating stable colloidal systems like hollandaise, and the principles of evaporation and flavour concentration in reduction sauces. Examiners award significant credit for the correct application of scientific terminology and a clear explanation of how ingredient function dictates the final product's characteristics. This guide will equip you with the detailed knowledge and exam technique required to confidently tackle questions on this topic and maximise your marks.
Key Scientific Principles
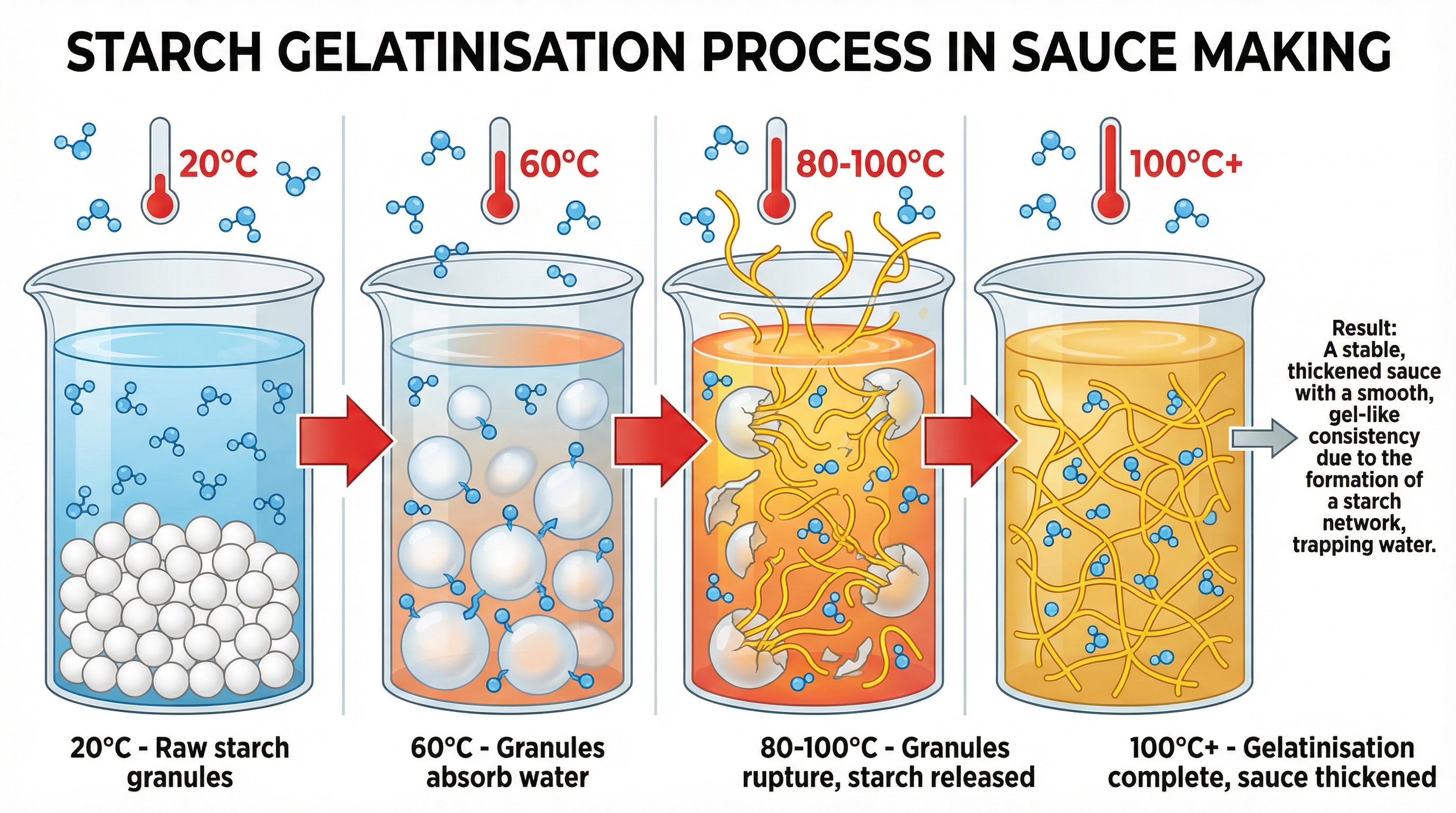
Gelatinisation (Roux-Based Sauces)
What happens: This process is fundamental to thickening sauces using starch, such as in a béchamel or velouté. When starch granules (from flour) are heated in a liquid (like milk or stock), they begin to absorb the liquid at around 60°C. As the temperature increases towards 100°C, the granules swell significantly, collide, and then rupture. This releases long-chain starch molecules (amylose and amylopectin) which unravel and form a complex network that traps water molecules.
Why it matters: This molecular network is what creates the thickened, viscous texture of the sauce. For full marks, candidates must explain this step-by-step process. It is essential to state that the sauce must reach boiling point (100°C) to ensure gelatinisation is complete and to cook out the raw taste of the flour. Dextrinisation (the browning of starch in a dark roux) can slightly reduce the thickening power of the starch, a nuance that can earn extra credit.
Specific Knowledge: Starch granules, swelling, rupture, viscosity, gel network, 100°C.
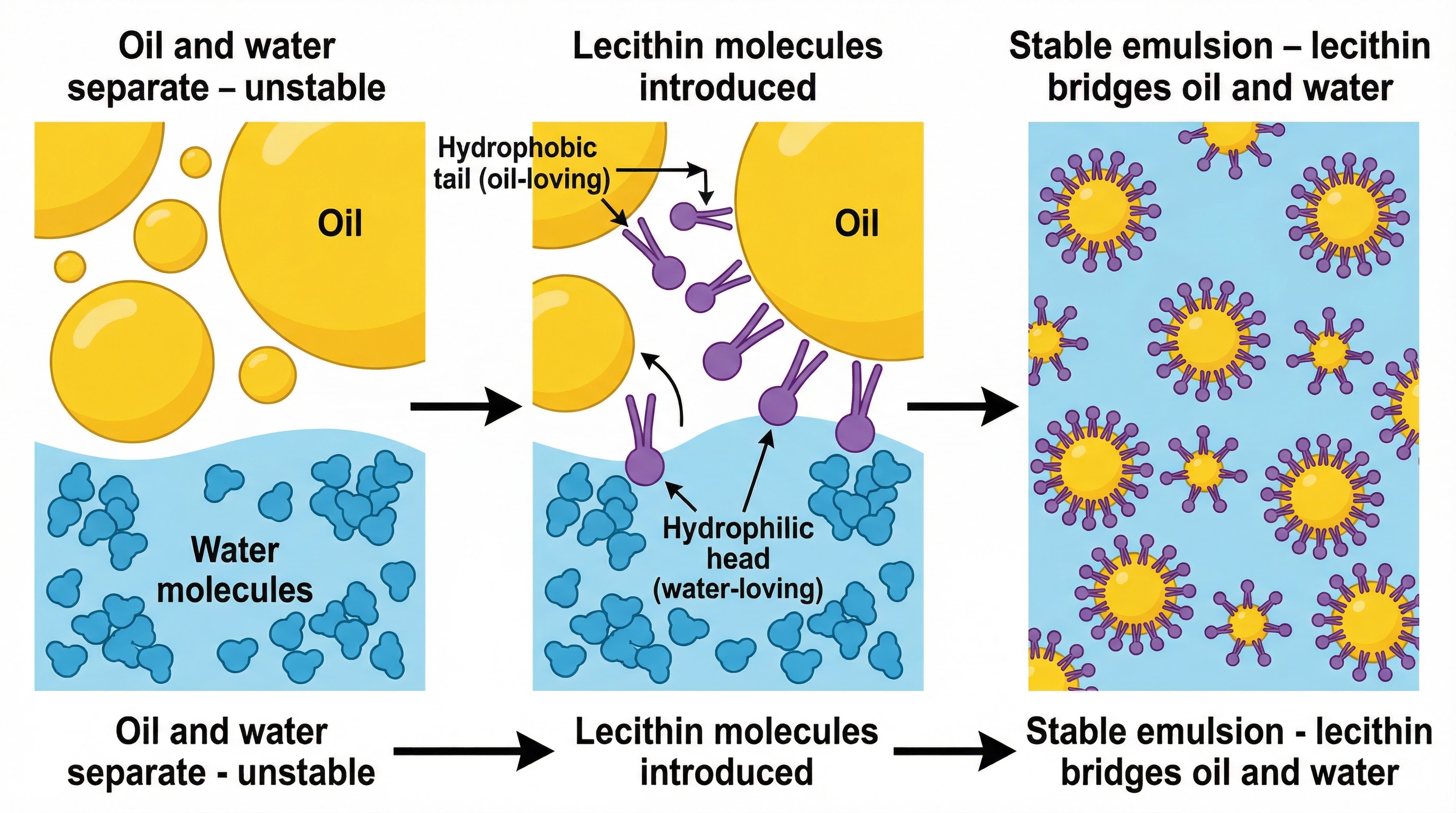
Emulsification (Colloidal Sauces)
What happened: Emulsification is the process of combining two immiscible liquids, like oil and water, into a stable mixture called a colloid. This is the principle behind sauces like hollandaise and mayonnaise. The key to this process is an emulsifier, a molecule that can bond with both liquids.
Why it matters: In hollandaise, the emulsifier is lecithin, found in egg yolk. Lecithin has a hydrophilic (water-loving) head and a hydrophobic (oil-loving) tail. When whisked, the lecithin molecules form a bridge between the water (from lemon juice) and the fat (from melted butter), creating tiny droplets of fat suspended in the water. This creates a stable, smooth, and thickened sauce. Candidates must explain this molecular bridge mechanism to gain full marks. Temperature control is critical; if the mixture exceeds 70°C, the egg proteins will coagulate, and the sauce will curdle.
Specific Knowledge: Lecithin, hydrophilic, hydrophobic, molecular bridge, colloid, coagulation, 65-70°C.
Reduction (Evaporation Sauces)
What happened: Reduction is the process of thickening a sauce and intensifying its flavour by simmering it to evaporate water content. This is common for tomato-based sauces, pan sauces, and wine reductions.
Why it matters: As water molecules turn to steam and leave the pan, the remaining ingredients—flavour compounds, sugars, acids, and proteins—become more concentrated. This leads to a richer taste and a naturally thicker consistency without the need for a separate thickening agent. Examiners expect candidates to differentiate between simmering (gentle, controlled evaporation at 85-95°C) and boiling (rapid, aggressive evaporation that can lead to burning and a loss of nuanced flavours).
Specific Knowledge: Evaporation, concentration, viscosity, simmering vs. boiling, 85-95°C.

Second-Order Concepts
Causation
- Gelatinisation: The application of heat causes starch granules to absorb liquid, swell, and rupture, which in turn causes the release of starch molecules that form a thickening network.
- Emulsification: The introduction of a shear force (whisking) and an emulsifying agent (lecithin) causes immiscible liquids (oil and water) to form a stable colloidal suspension.
- Reduction: The sustained application of gentle heat (simmering) causes water to evaporate, which causes the flavours in the remaining liquid to concentrate and the viscosity to increase.
Consequence
- Gelatinisation: The consequence is a stable, thickened sauce with a smooth texture, but if undercooked, it will have a raw flour taste. Overheating can cause the starch network to break down, thinning the sauce.
- Emulsification: The consequence is a smooth, stable, and rich-tasting sauce. However, incorrect temperature control leads to coagulation and a split (curdled) sauce.
- Reduction: The consequence is a sauce with a deep, complex flavour profile and a naturally thick consistency. However, reducing too far can make the sauce overly salty or acidic.
Change & Continuity
- Change: The physical state of the ingredients changes dramatically. Raw, granular starch becomes a smooth gel. Separate liquids become a unified emulsion. A thin liquid becomes a viscous, concentrated sauce.
- Continuity: The chemical nature of the core ingredients remains. The starch molecules are still starch, just rearranged. The oil and water are still present, just held in suspension.
Significance
- The significance of understanding these scientific principles is that it allows a chef to manipulate ingredients to create a desired texture and flavour, moving beyond a simple recipe to control the outcome. For an exam candidate, it is the key to unlocking AO2 marks for application and analysis."