Study Notes

Overview
Absolute Zero is a cornerstone of thermodynamics, representing the theoretical lowest possible temperature. For your OCR GCSE Physics exam, a solid understanding of this topic is crucial for questions related to the particle model of matter and gas laws. This guide will break down the essential knowledge, from the definition of Absolute Zero (0 Kelvin or -273°C) to its implications for particle motion. We will explore the direct relationship between the temperature of a gas and its pressure at a constant volume, a concept frequently tested through calculations and graph interpretation. Expect to see questions that require you to convert between Celsius and Kelvin, analyse pressure-temperature graphs, and apply the kinetic theory to explain gas behaviour. Mastering these skills will not only secure marks in this section but also strengthen your understanding of energy and matter across the physics specification.
Key Concepts
Concept 1: Absolute Zero and the Kelvin Scale
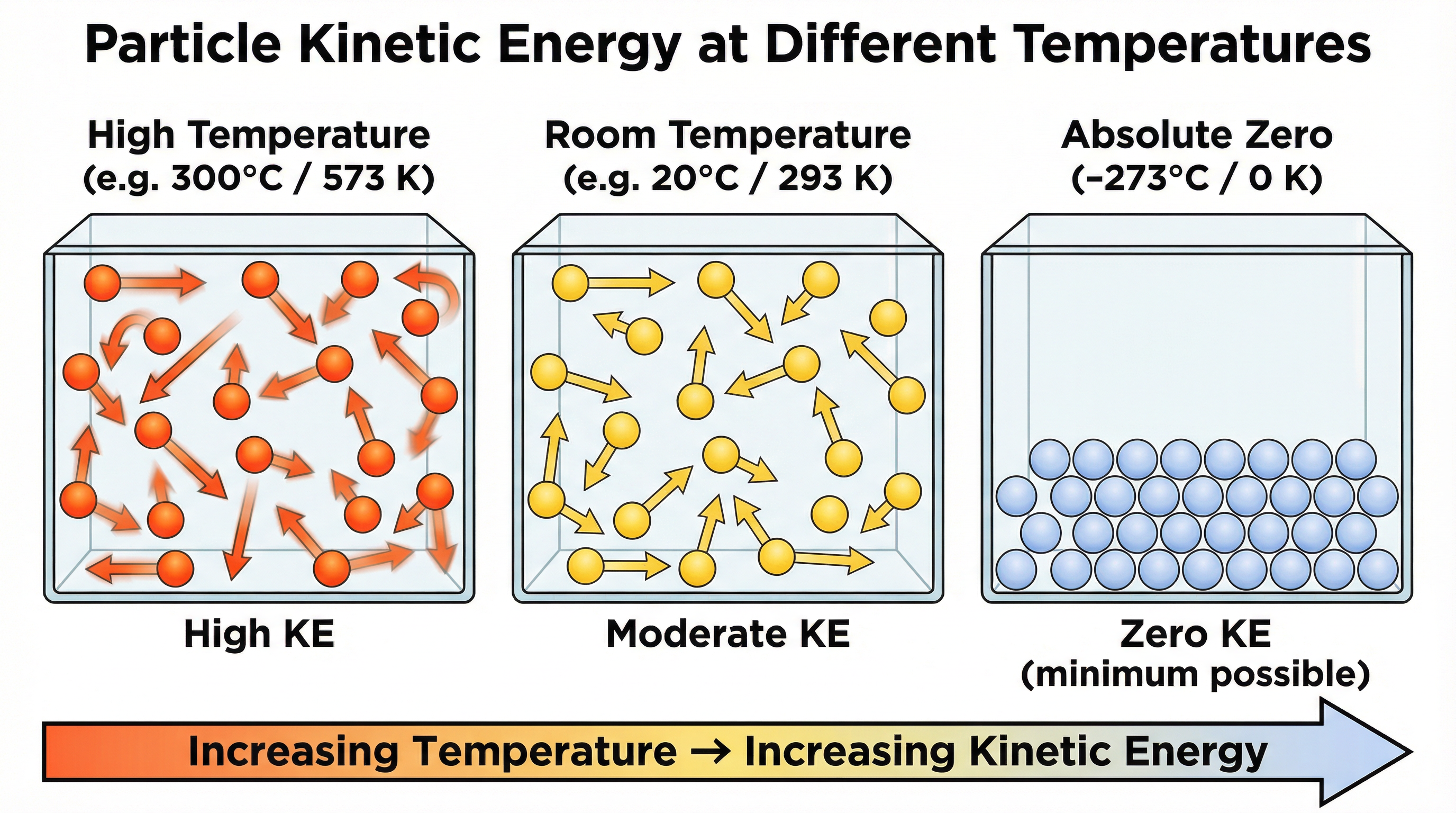
Absolute Zero is the point at which particles have their minimum possible kinetic energy. It is the coldest temperature that can be theoretically reached. On the Celsius scale, this is -273 °C. Physicists use the Kelvin scale for thermodynamic calculations because it is an absolute scale, meaning 0 K is absolute zero. This avoids the negative numbers and makes relationships, like the one between pressure and temperature, much simpler to express.
Example: To convert a room temperature of 20 °C to Kelvin, you add 273. So, 20 + 273 = 293 K. This conversion is a fundamental skill that you will be expected to apply in exam questions.
Concept 2: Kinetic Theory of Gases
The kinetic theory explains the macroscopic properties of gases (like pressure and temperature) by considering the motion of their constituent particles. The theory is based on three main assumptions:
- The gas consists of a large number of identical particles (atoms or molecules).
- The particles are in constant, random motion.
- The collisions between particles and with the walls of the container are perfectly elastic (no kinetic energy is lost).
Temperature is a measure of the average kinetic energy of the particles. The higher the temperature, the higher the average kinetic energy, and the faster the particles move.
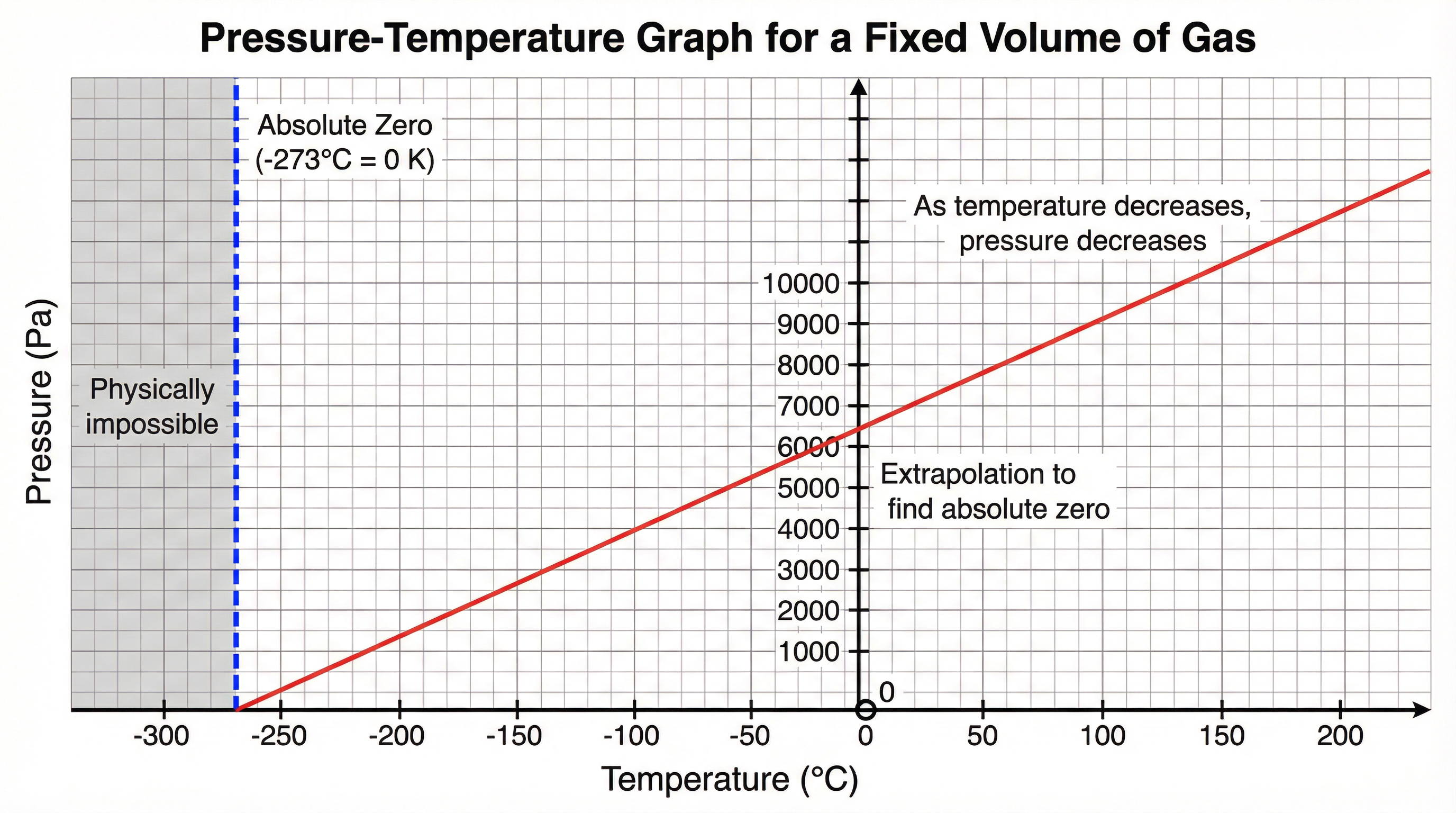
Concept 3: The Pressure-Temperature Relationship
For a fixed mass of gas at a constant volume, the pressure of the gas is directly proportional to its absolute temperature in Kelvin. This is known as Gay-Lussac's Law or the Pressure Law. This means if you double the absolute temperature (in Kelvin), you double the pressure. This relationship is explained by the kinetic theory: as temperature increases, particles move faster and collide with the container walls more frequently and with greater force, increasing the pressure.
Mathematical/Scientific Relationships
-
Celsius to Kelvin Conversion (Must memorise):
T(K) = T(°C) + 273 -
Kelvin to Celsius Conversion (Must memorise):
T(°C) = T(K) - 273 -
The Pressure Law (Given on formula sheet):
P₁/T₁ = P₂/T₂
Where:P₁is the initial pressureT₁is the initial absolute temperature (in Kelvin)P₂is the final pressureT₂is the final absolute temperature (in Kelvin)
Practical Applications
While reaching absolute zero is practically impossible, the principles are fundamental to many technologies. Cryogenics, the study of very low temperatures, is used for:
- Superconducting Magnets: Used in MRI scanners and particle accelerators.
- Food Preservation: Freezing food at very low temperatures slows down the process of decay.
- Rocket Fuel: Liquefied gases like hydrogen and oxygen are used as rocket propellants.