Study Notes

Overview
Isotopes are a fundamental concept in atomic structure, forming a cornerstone of your OCR GCSE Physics course. This topic explores why atoms of the same element can have different masses and how we represent this using standard scientific notation. A solid understanding of isotopes is not just about memorising definitions; it's about applying that knowledge to calculate the number of particles in an atom and linking it to bigger ideas like nuclear stability and radioactivity, which are covered later in the course. Examiners frequently test this area with a mix of definition-based questions (AO1) and application-based problems (AO2), so precision is key. Expect to see questions asking you to define isotopes, calculate neutron numbers, or compare the subatomic structures of different atoms.
Key Concepts
Concept 1: The Definition of an Isotope
In simple terms, isotopes are different versions of the same element. For a definition that will earn you marks in an exam, you must be more precise. Examiners at OCR are looking for a specific two-part answer:
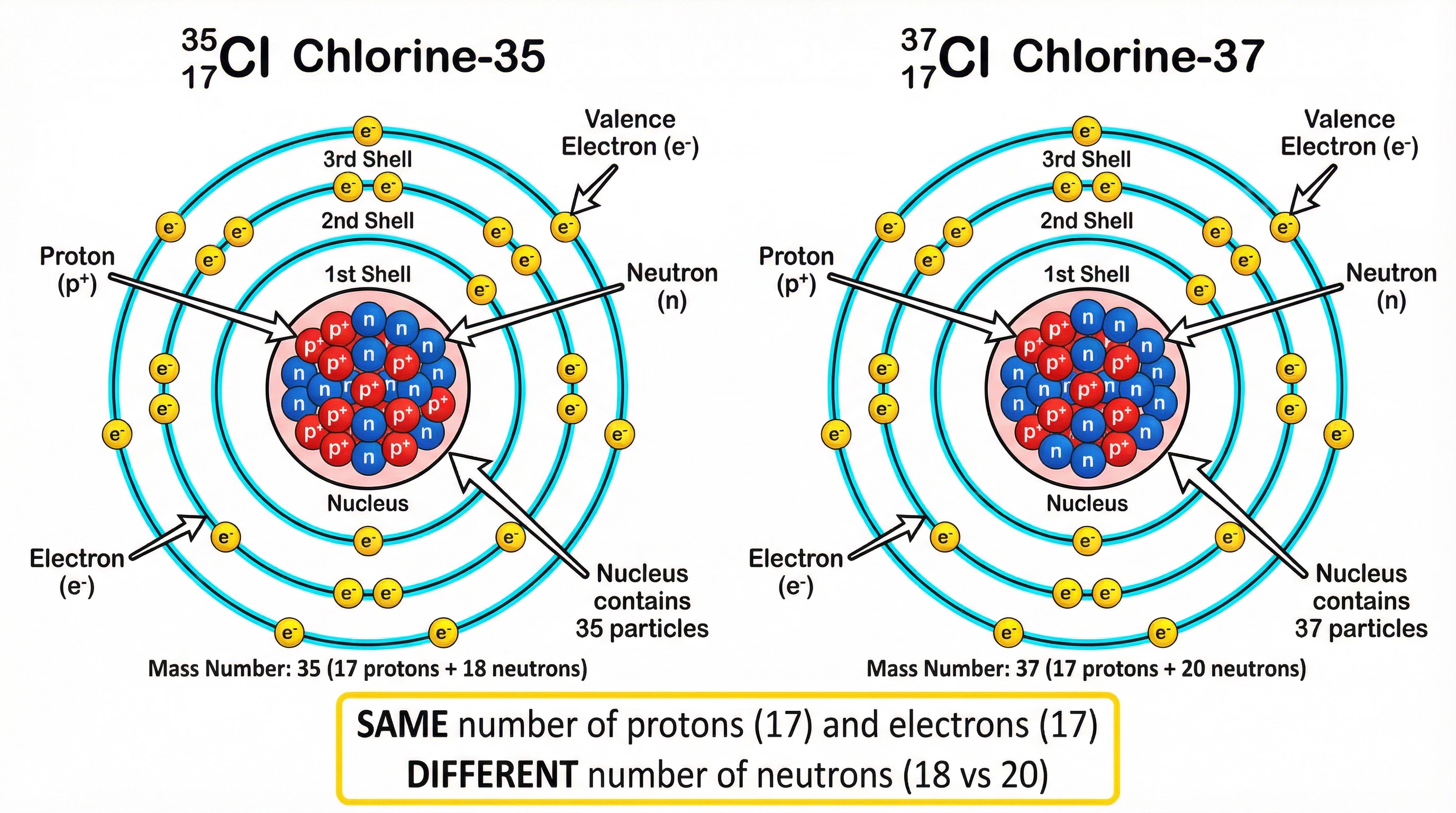
**Isotopes are atoms of the same element that have the same number of protons but a different number of neutrons.**Let's break that down. The number of protons in an atom's nucleus determines what element it is. This is its atomic number (Z). Every single carbon atom in the universe has 6 protons. If it had 7, it would be nitrogen. This part is non-negotiable. However, the number of neutrons can vary. Adding or removing neutrons from the nucleus doesn't change the element, but it does change the atom's mass. This creates different isotopes of that element.
Example: All carbon atoms have 6 protons.
- Carbon-12 has 6 protons and 6 neutrons.
- Carbon-13 has 6 protons and 7 neutrons.
- Carbon-14 has 6 protons and 8 neutrons.
They are all isotopes of carbon because the proton number is the same, but the neutron number is different.
Concept 2: Subatomic Particles and Nuclear Notation
To work with isotopes, you need to be fluent in the language of subatomic particles and the standard notation used to represent them. An atom is made of three key particles:
| Particle | Relative Charge | Relative Mass | Location in Atom |
|---|---|---|---|
| Proton | +1 | 1 | Nucleus |
| Neutron | 0 | 1 | Nucleus |
| Electron | -1 | ~1/2000 (negligible) | Shells/Orbitals |
Nuclear notation (or nuclide notation) is the standard way to write down the composition of a nucleus. It looks like this:
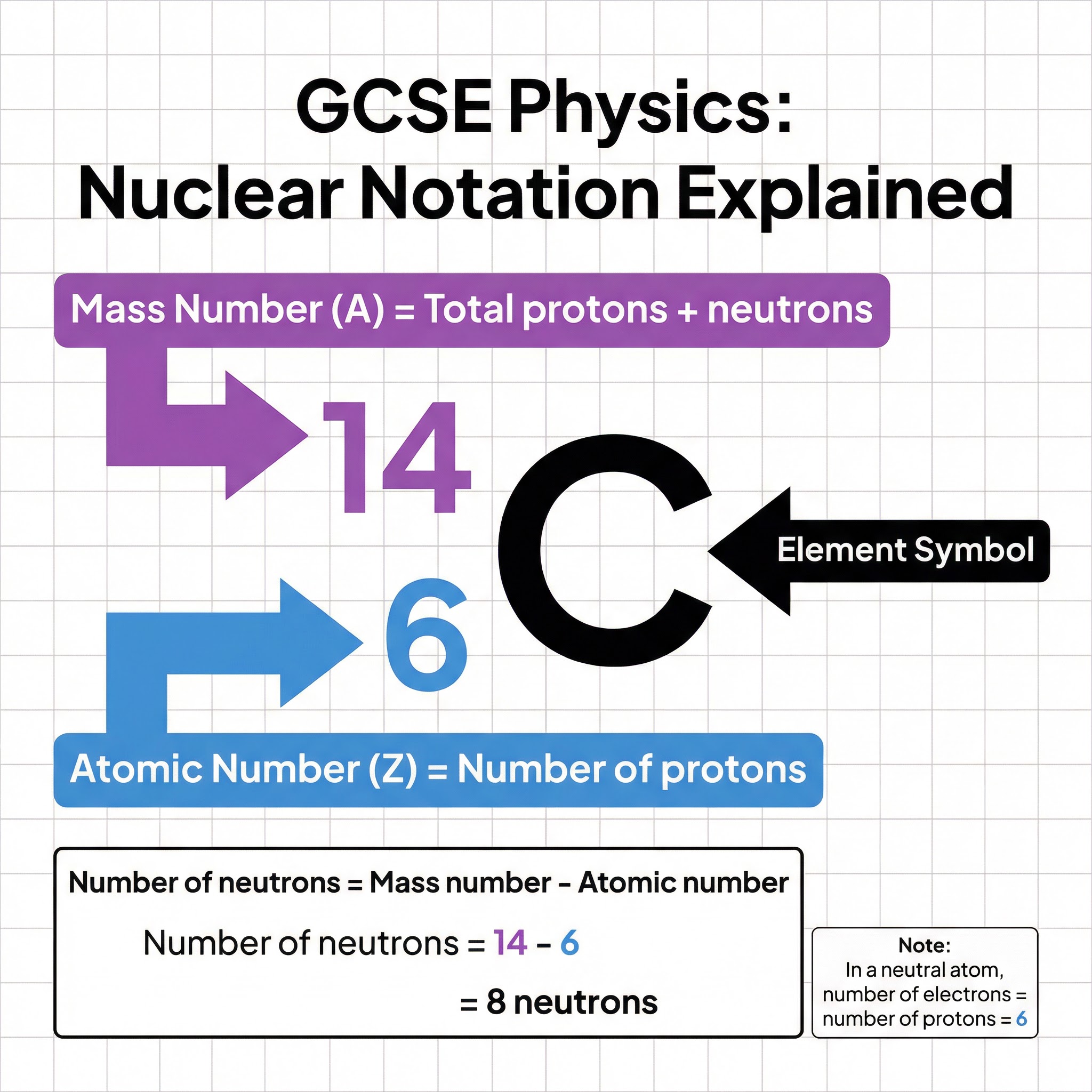
- A is the Mass Number: The total number of protons and neutrons in the nucleus.
- Z is the Atomic Number: The number of protons in the nucleus.
- X is the Element Symbol: The one or two-letter symbol for the element (e.g., C for Carbon).
Crucially, in a neutral atom, the number of electrons is equal to the number of protons. This is why isotopes of the same element have identical chemical properties – their electron structure is the same, and it's the electrons that determine how an atom reacts.
Concept 3: Calculating Subatomic Particles
Examiners will expect you to be able to calculate the number of protons, neutrons, and electrons for any given isotope. It's a simple process if you understand the notation.
- Number of Protons = Atomic Number (Z)
- Number of Electrons = Atomic Number (Z) (for a neutral atom)
- Number of Neutrons = Mass Number (A) - Atomic Number (Z)
Example: Let's look at an isotope of uranium, Uranium-235. The atomic number is 92 and the mass number is 235.
- Protons: The atomic number is 92. So, there are 92 protons.
- Electrons: It's a neutral atom, so the number of electrons equals the number of protons. There are 92 electrons.
- Neutrons: The mass number is 235. Number of neutrons = 235 - 92 = 143 neutrons.
Mathematical/Scientific Relationships
The key formula you must know is for calculating the number of neutrons. It is not typically given on the formula sheet, so you must memorise it.
- Formula:
Number of Neutrons = Mass Number (A) - Atomic Number (Z)- A (Mass Number): Total particles in the nucleus. Must be taken from the specific isotope in the question (e.g., the '14' in Carbon-14).
- Z (Atomic Number): Number of protons. Defines the element.
Another important concept is Relative Atomic Mass (Ar). This is the value shown on the Periodic Table. It is a weighted average mass of all the isotopes of an element, taking into account their relative abundances. For example, chlorine has two main isotopes, Chlorine-35 (75% abundance) and Chlorine-37 (25% abundance). Its relative atomic mass is calculated as:
Ar = ( (75/100) * 35 ) + ( (25/100) * 37 ) = 26.25 + 9.25 = 35.5
This is a Higher Tier concept, but it helps explain why the mass numbers on the periodic table are often not whole numbers.
Practical Applications
Isotopes aren't just an abstract concept; they have vital real-world applications.
- Carbon Dating: The radioactive isotope Carbon-14 is used to determine the age of organic materials. Living things absorb carbon from the atmosphere. Once they die, the Carbon-14 decays at a known rate (its half-life). By measuring the remaining Carbon-14, scientists can calculate how long ago the organism died. This is a synoptic link to the topic of Radioactivity.
- Medical Tracers: Radioactive isotopes can be injected into the body to monitor internal processes. For example, Iodine-131 is used to check thyroid function because the thyroid gland absorbs iodine. A detector outside the body tracks the gamma rays emitted by the tracer, creating an image of the organ. The isotopes used must have a short half-life to minimise radiation dose to the patient.
- Nuclear Power: Uranium-235 is a fissile isotope used as fuel in nuclear power stations. Its nucleus can be split apart, releasing huge amounts of energy. Uranium-238, the more common isotope, does not split in this way.