Study Notes

Overview
Atmospheric pressure is a fundamental concept in physics, describing the force exerted by the weight of the air in the atmosphere. For your OCR GCSE Physics exam, you need to understand this topic on two levels: the macroscopic (the weight of the entire air column above you) and the microscopic (the constant bombardment of air molecules). This topic is crucial as it links to ideas about forces, pressure, and the particle model of matter. Examiners frequently test this with graph interpretation questions and multi-mark explanations, so a precise understanding is essential for achieving higher grades. This guide will break down the core concepts, provide worked examples, and give you the exam technique needed to answer any question on atmospheric pressure with confidence.
Key Concepts
Concept 1: The Two Models of Atmospheric Pressure
There are two ways to conceptualise atmospheric pressure, and examiners expect you to be familiar with both.
-
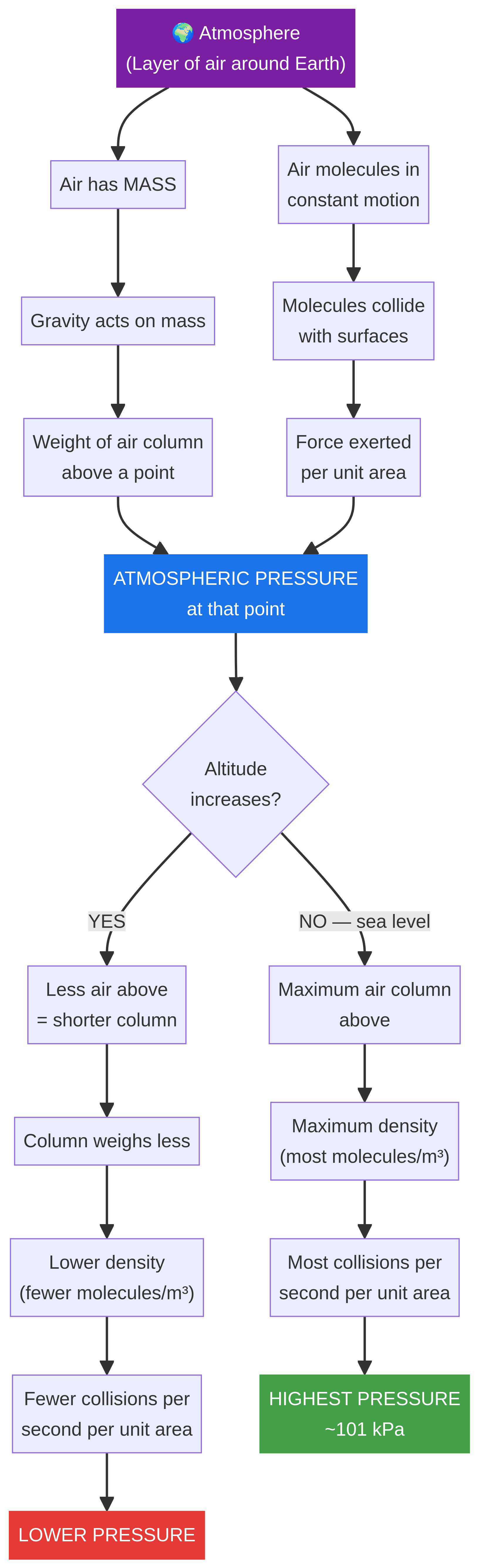
The Macroscopic Model (Weight of Air): The simplest way to think about it is that the atmosphere is a huge ocean of air. Air, although it seems weightless, has mass. Gravity pulls this mass towards the Earth. Therefore, the air above you exerts a force on the area beneath it. This force per unit area is what we call atmospheric pressure. Imagine a 1m² column of air stretching from sea level to the edge of space – it would weigh over 100,000 Newtons!
-
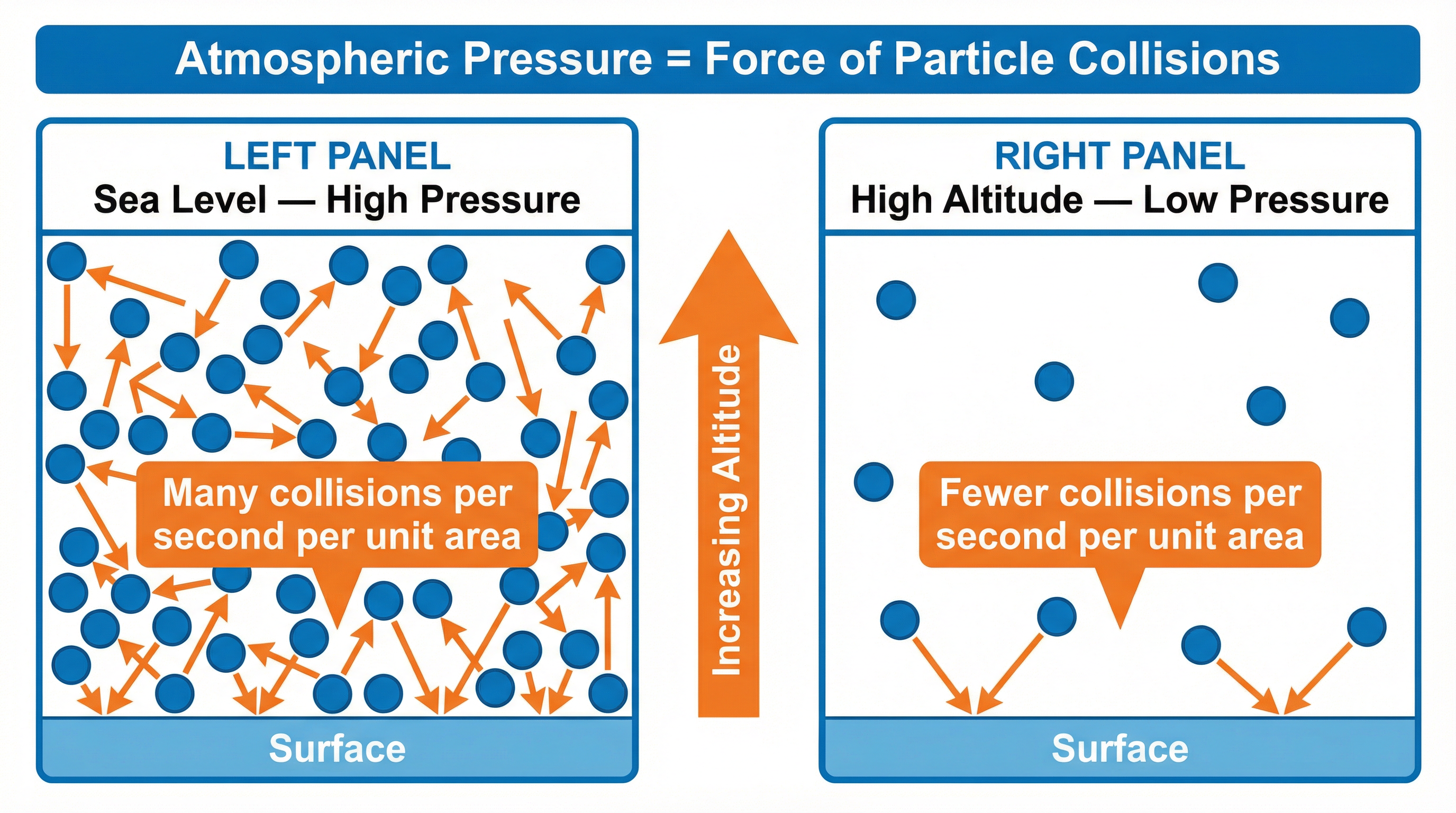
The Microscopic Model (Particle Collisions): This is the more detailed physical explanation. The air is composed of billions of tiny, fast-moving particles (molecules). These particles are in constant, random motion, and they collide with every surface they come into contact with. Each collision exerts a tiny force. Atmospheric pressure is the total force of all these collisions added up over a certain area. This is the explanation that unlocks the highest marks in an exam.
Concept 2: Altitude and its Effect on Pressure
This is the most commonly examined part of the topic. As altitude increases, atmospheric pressure decreases. You must be able to explain WHY this happens using the particle model.
- Decreasing Density: As you go higher, the density of the air decreases. This is because there is less air above pushing down, so the particles are more spread out. The correct term is that the air is less dense, meaning there are fewer air molecules per unit volume (e.g., per cubic metre).
- Reduced Collision Frequency: Because there are fewer molecules in a given volume at higher altitudes, there will be fewer collisions with any surface per second. It is this reduction in the frequency of collisions that leads to a lower average force on the surface, and therefore, lower pressure.
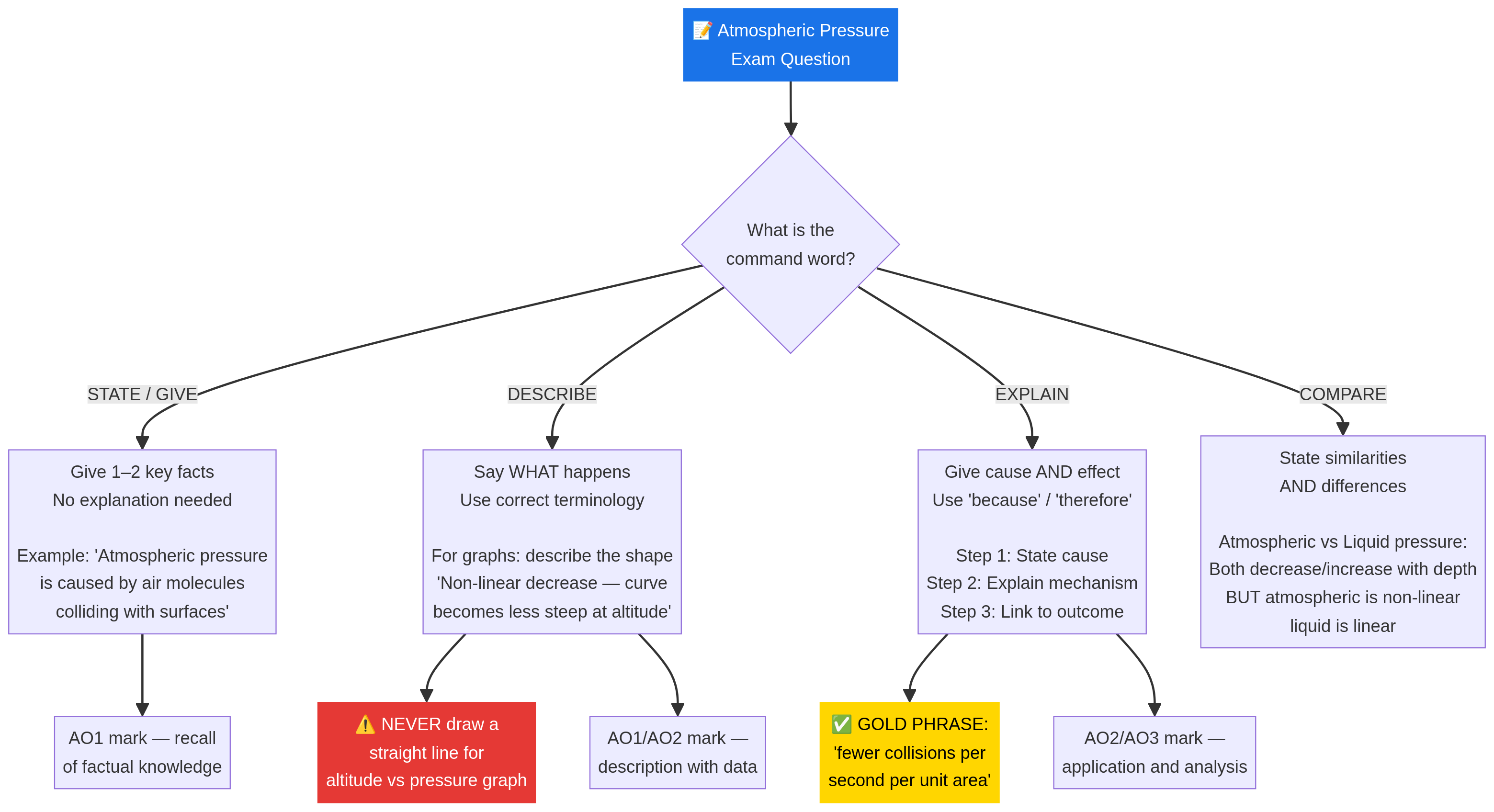
Examiner Tip: The phrase ‘fewer collisions per second per unit area’ is a high-scoring phrase that you should aim to use in your explanations.
Concept 3: The Non-Linear Relationship
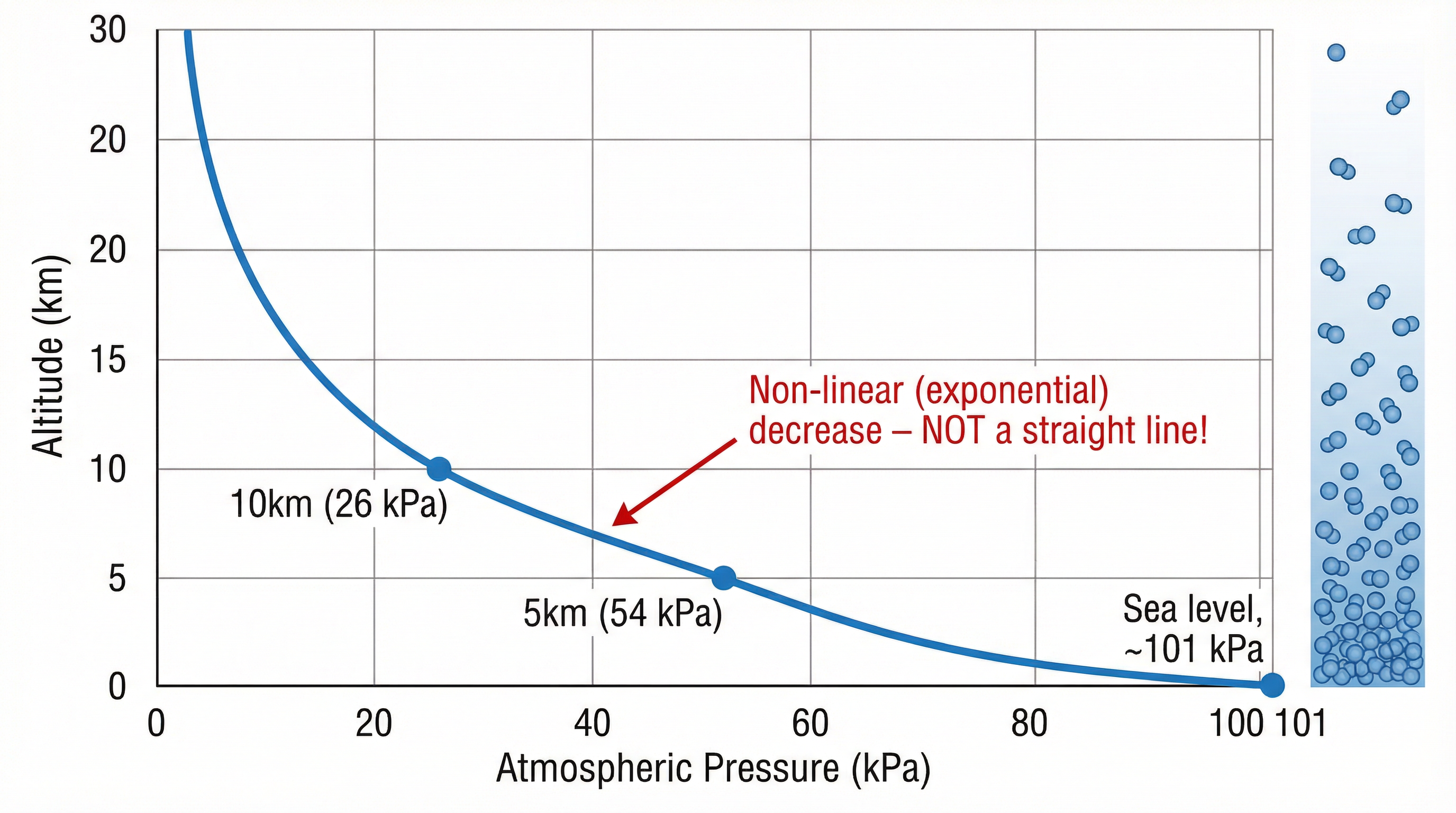
Unlike pressure in a liquid (which increases linearly with depth), the relationship between atmospheric pressure and altitude is non-linear. This is a crucial distinction.
- Why it's a curve: The pressure does not decrease by the same amount for every 1000m you ascend. The drop is sharpest near sea level where the air is densest, and becomes more gradual at higher altitudes where the air is already very thin. This is because the air itself is compressible. The lower layers are squashed by the weight of the layers above, making them denser. As you go up, the density decreases, so the rate of pressure change also decreases.
- Graph Interpretation: In an exam, if you are asked to draw or interpret a graph of pressure vs. altitude, it MUST be a curve that gets less steep as altitude increases. Drawing a straight line is a common mistake that will lose you marks.
Mathematical/Scientific Relationships
There are no complex formulas you need to memorise for atmospheric pressure at GCSE level, unlike the formula for pressure in liquids (P = hρg). The key relationship is conceptual:
- Pressure ∝ 1 / Altitude (Non-linearly): As altitude increases, pressure decreases, but not in a simple proportional way. It's an exponential decay relationship.
- Pressure ∝ Density of Air: Where the air is denser, the pressure is higher.
- Pressure ∝ Frequency of Particle Collisions: More collisions per second per unit area result in higher pressure.
Practical Applications
- Weather Forecasting: Barometers measure atmospheric pressure. A falling pressure often indicates an approaching low-pressure system, which typically brings unsettled weather (clouds, rain). Rising pressure suggests a high-pressure system, associated with clear skies and calm conditions.
- Aviation: Altimeters in aircraft are essentially barometers. By measuring the outside air pressure, they can calculate the aircraft's altitude above sea level.
- Drinking with a Straw: When you suck on a straw, you reduce the air pressure inside it. The higher atmospheric pressure outside then pushes the liquid up the straw and into your mouth.