Study Notes
Overview
The Electromagnetic (EM) Spectrum is a continuous range of waves that are fundamental to our understanding of the universe and essential to modern technology. For an OCR GCSE Physics candidate, mastering this topic is non-negotiable. It bridges concepts from waves, atomic structure, and space physics, making it a prime candidate for synoptic questions. Examiners expect you to not only recall the order and properties of the seven distinct wave types but also to apply this knowledge to real-world contexts, from satellite communication to medical imaging. You will be tested on your ability to describe how these waves are generated and absorbed, explain their uses and associated hazards, and perform calculations using the wave equation. A solid grasp of the inverse relationship between frequency and wavelength is crucial for achieving the highest marks.
Key Concepts
Concept 1: The Universal Properties of EM Waves
All electromagnetic waves, regardless of their type, share two defining characteristics that you must be able to state for exam credit. Firstly, they are all transverse waves. This means the oscillations (vibrations) of the electric and magnetic fields are perpendicular (at 90 degrees) to the direction of energy transfer. Secondly, and this is a critical marking point, all EM waves travel at the same speed in a vacuum: 3.0 x 10⁸ m/s. This value, known as the speed of light (c), is a constant. It is a common mistake for candidates to think that gamma rays travel faster than radio waves; they do not. This constant speed is the foundation for the core mathematical relationship in this topic.
Concept 2: The Inverse Relationship - Frequency and Wavelength
The EM spectrum is arranged in a specific, continuous order based on the waves' frequency and wavelength. As you move along the spectrum from radio waves to gamma rays, the frequency increases while the wavelength decreases. This is an inverse relationship. Because the wave speed (v) is constant, if the frequency (f) of a wave increases, its wavelength (λ) must decrease proportionally to maintain the relationship defined by the wave equation.
Concept 3: The Seven Regions of the Spectrum
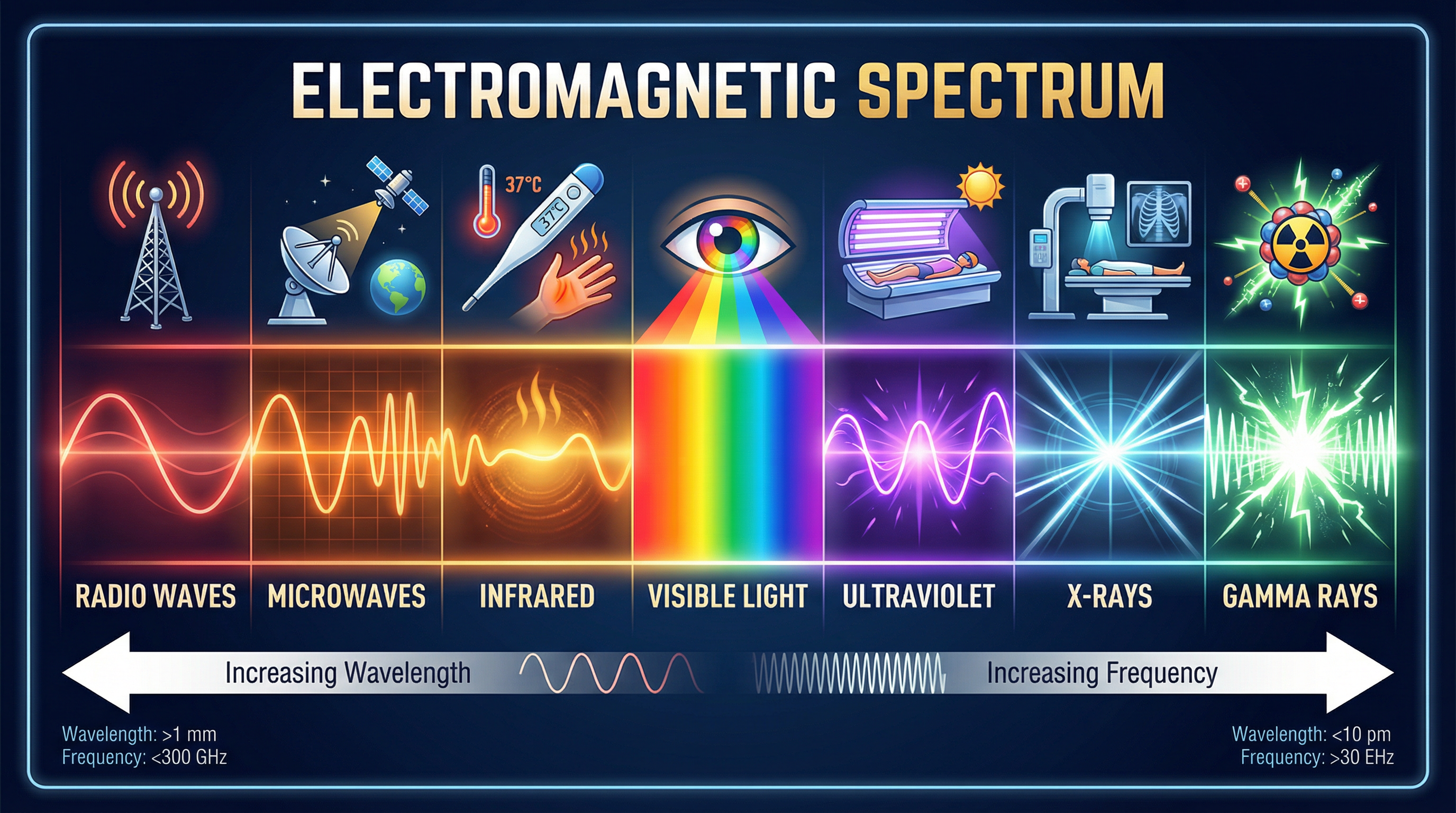
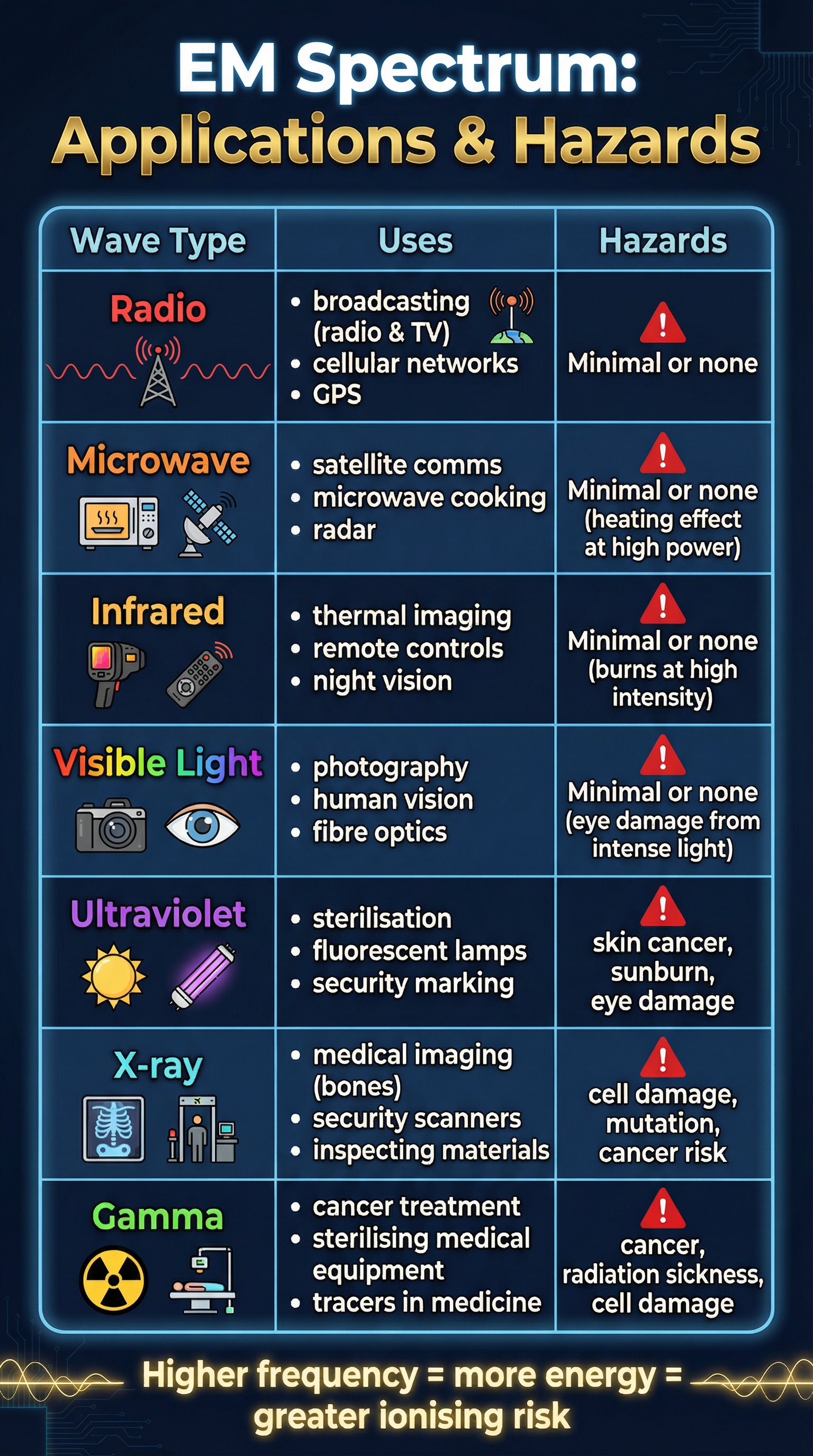
Candidates must be able to recall the order of the seven types of EM waves and provide uses and dangers for each. The order is: Radio waves, Microwaves, Infrared, Visible light, Ultraviolet, X-rays, Gamma rays.
- Radio waves: Longest wavelength, lowest frequency. Used for broadcasting TV and radio signals. (Higher Tier: Produced by oscillations in electrical circuits).
- Microwaves: Used for satellite communications and heating food. The key mechanism for heating is that microwaves are absorbed by water molecules, causing them to vibrate and transfer thermal energy to the rest of the food.
- Infrared (IR): Emitted by all objects. Used in thermal imaging cameras (e.g., for night vision) and remote controls.
- Visible Light: The only part of the spectrum detectable by the human eye. Used in photography and fibre optics.
- Ultraviolet (UV): Used in fluorescent lamps and for sterilising water. It is the first of the high-energy, ionising waves.
- X-rays: Used in medical imaging to see bones and in airport security scanners. They can penetrate soft tissue but are absorbed by denser materials like bone.
- Gamma rays: Highest frequency, shortest wavelength. Used to sterilise medical equipment and in radiotherapy to treat cancer. (Higher Tier: Emitted from the nucleus of an atom during radioactive decay).
Concept 4: Ionising Radiation
The high-frequency end of the spectrum – Ultraviolet, X-rays, and Gamma rays – carries enough energy in each photon to be classified as ionising radiation. This means they have sufficient energy to knock electrons from atoms, creating ions. This process can damage or kill living cells, leading to an increased risk of mutations and cancer. The higher the frequency, the more ionising the radiation. This is why exposure to these types of radiation is carefully controlled.
Mathematical/Scientific Relationships
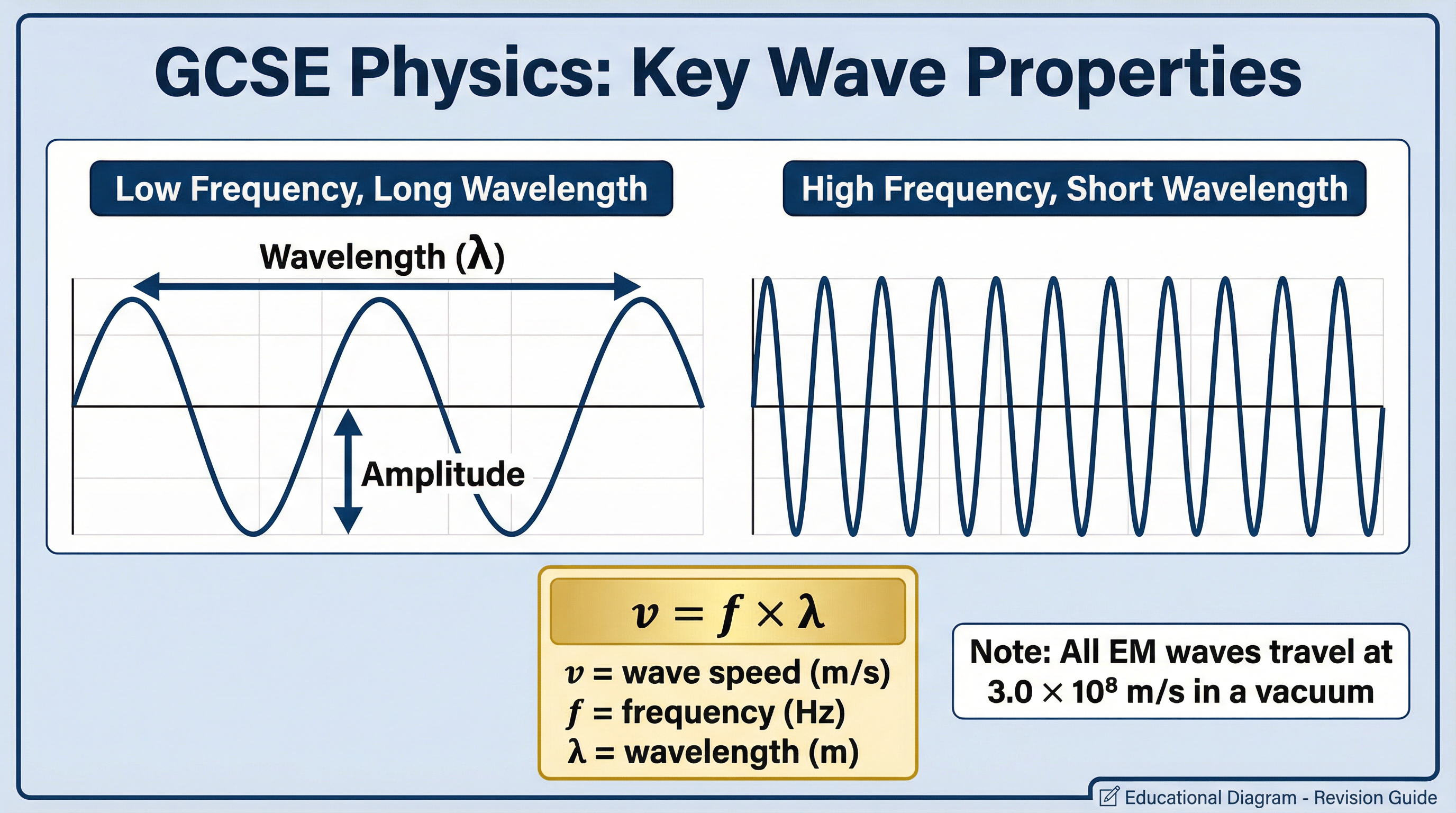
The Wave Equation (Must memorise)
The fundamental formula linking the properties of any wave is the wave equation:
v = f × λWhere:
- v is the wave speed, measured in metres per second (m/s). For all EM waves in a vacuum, this is 3.0 x 10⁸ m/s.
- f is the frequency, measured in Hertz (Hz).
- λ (lambda) is the wavelength, measured in metres (m).
Examiners will expect you to recall and apply this formula. You may be asked to calculate any of the three variables, given the other two. Remember to convert all units to their standard forms (e.g., kHz to Hz, cm to m) before substituting them into the equation.
Practical Applications
This topic is rich with real-world applications that are frequently tested.
- Communications: Radio waves are used for long-range TV and radio transmission. Microwaves are used for satellite TV and mobile phone calls because they can pass through the Earth's atmosphere without being significantly reflected or refracted.
- Medical Imaging: X-rays are invaluable for diagnosing bone fractures because of their differential absorption by bone and soft tissue. Gamma rays can be used as tracers in medical imaging, where a patient ingests a weak gamma source and its path through the body is tracked.
- Cancer Treatment: Because gamma rays are highly ionising and can kill cells, a concentrated beam can be directed at a cancerous tumour to destroy it. This is known as radiotherapy.
- Heating and Cooking: Microwaves heat food by causing water molecules to vibrate. Infrared heaters and grills transfer thermal energy directly to the surface of food.