Study Notes

Overview
Welcome to your definitive guide for OCR GCSE Physics Topic 6.7: Gas Pressure. This topic is fundamental, explaining the behaviour of gases from a microscopic and macroscopic perspective. Examiners frequently test this area because it links several key physics principles: forces, energy, and the particle model of matter. You will learn how countless tiny, fast-moving particles create the pressure that inflates a tyre or pushes a piston. We will explore the crucial relationship between pressure and volume (Boyle's Law) and, for Higher Tier candidates, the link between doing work on a gas and its internal energy. Expect to see a mix of short-answer explanation questions (worth 1-3 marks) and longer calculation or extended-response questions (worth 4-6 marks). Mastering the concepts and, more importantly, the specific language required by the mark scheme is the key to success here.
Key Concepts
Concept 1: The Particle Model of Gas Pressure
At the heart of this topic is the idea that a gas consists of a large number of particles (atoms or molecules) that are in constant, random motion. They move at high speeds and in random directions. Gas pressure is the result of these particles colliding with the walls of their container. Each collision exerts a small force on the wall. When you sum up the total force of all these collisions over the area of the container walls, you get the pressure. For a stable pressure, the rate of collisions must be constant.
Examiner's Tip: To earn the marks for explaining the origin of gas pressure, you must state that particles collide with the walls of the container. A common mistake that gets zero credit is saying particles collide with 'each other'. While they do, this does not cause the pressure on the container.
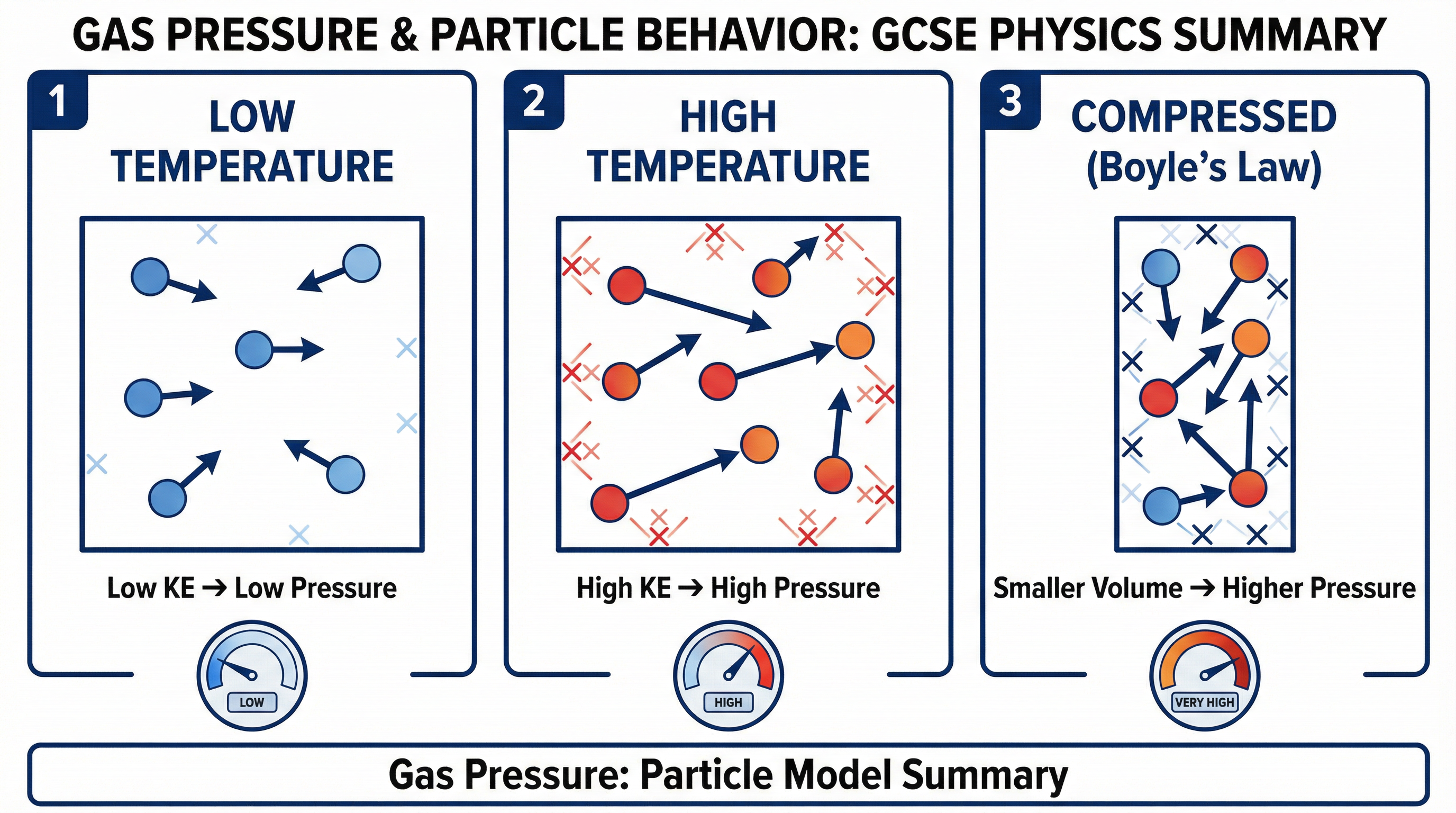
Concept 2: The Relationship Between Temperature and Pressure
If you heat a gas in a fixed-volume container, its pressure increases. This is because heating the gas transfers thermal energy to its particles, increasing their internal energy store. Specifically, the kinetic energy of the particles increases. This means they move faster. Faster-moving particles lead to two effects:
- More Frequent Collisions: The particles hit the container walls more often per unit of time.
- More Forceful Collisions: Each collision involves a greater change in momentum, resulting in a larger force.
Both factors increase the total force exerted on the walls, and therefore the pressure increases. For a fixed volume, the pressure of a gas is directly proportional to its absolute temperature (measured in Kelvin). This is known as the Pressure Law.
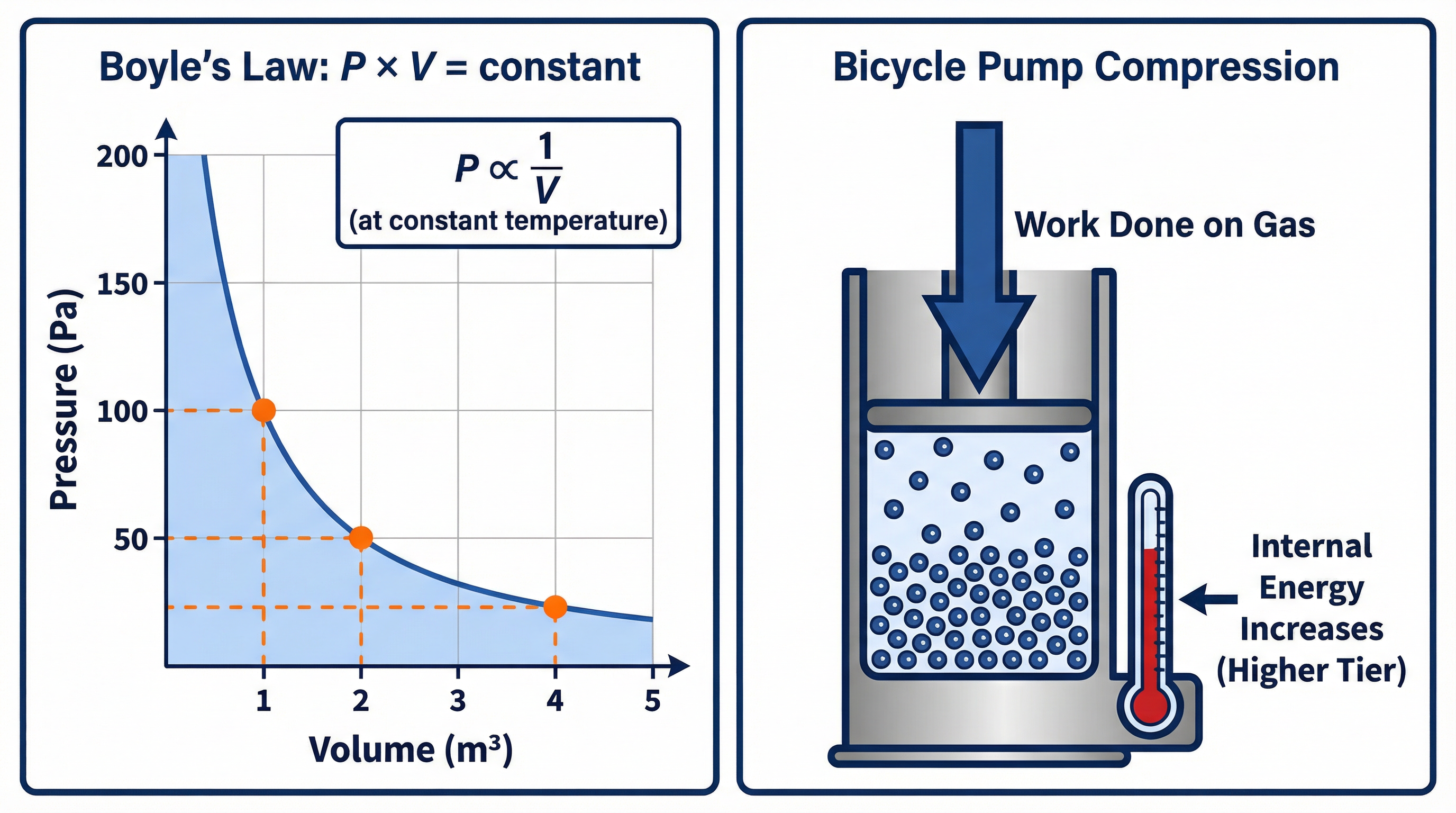
Concept 3: Boyle's Law - The Relationship Between Pressure and Volume
For a fixed mass of gas at a constant temperature, the pressure and volume are inversely proportional. This is Boyle's Law. This means if you double the volume, you halve the pressure. If you decrease the volume, the pressure increases. Why? If you compress a gas into a smaller volume, the particles are more crowded. They have less distance to travel before hitting a wall, so the frequency of collisions with the walls increases. More frequent collisions per unit area result in a higher pressure.
Mathematical/Scientific Relationships
Here are the key formulas you need to know for this topic. Pay close attention to which ones are provided and which you must memorise.
-
Pressure, Force, Area
- Formula:
P = F / A - Symbols: P = Pressure (in Pascals, Pa), F = Force (in Newtons, N), A = Area (in metres squared, m²)
- Status: Must memorise.
- Notes: This is a fundamental definition. Be very careful with area conversions; examiners often give areas in cm² to catch candidates out. 1 m² = 10,000 cm².
- Formula:
-
Boyle's Law
- Formula:
P₁V₁ = P₂V₂ - Symbols: P₁ = Initial Pressure, V₁ = Initial Volume, P₂ = Final Pressure, V₂ = Final Volume.
- Status: Given on formula sheet.
- Notes: This formula is used to calculate the change in pressure or volume of a fixed mass of gas at constant temperature. The units for pressure and volume must be consistent on both sides of the equation (e.g., if P₁ is in kPa, P₂ will be in kPa; if V₁ is in cm³, V₂ will be in cm³). You don't necessarily need to convert to standard SI units, as long as you are consistent.
- Formula:
Practical Applications
The Bicycle Pump (Required Practical Context)
This is a classic context used by OCR. When you push the handle of a bicycle pump, you are doing mechanical work on the gas (air) trapped inside. This work transfers energy to the gas particles, increasing their internal energy. This leads to an increase in the gas's temperature. This is a key concept for Higher Tier candidates. You can feel the pump getting warm as you use it. This is not due to friction alone, but primarily because you are doing work on the gas.
For a 6-mark question on this, a good answer would link the following chain of reasoning:
- A force is applied to the pump handle, which does work on the piston.
- This work is done on the gas inside the pump, transferring energy to the gas particles.
- The internal energy of the gas increases, meaning the average kinetic energy of the particles increases.
- An increase in the average kinetic energy of the particles is equivalent to an increase in the temperature of the gas.
- Simultaneously, the volume is decreasing, which increases the frequency of particle collisions with the walls, further increasing pressure.
Other Applications
- Aerosol Cans: These come with warnings not to incinerate them. Heating the can increases the pressure of the gas inside, which could cause it to explode.
- Weather Systems: Large-scale changes in atmospheric pressure drive our weather patterns.
- Diving: As divers go deeper, the water pressure increases, which affects the volume of gases in their bodies and equipment.