Study Notes

Overview
Internal energy is a fundamental concept in thermodynamics and the particle model of matter. For your OCR GCSE Physics exam, it is defined with mark-winning precision as the total kinetic and potential energy of all the particles that make up a system. Understanding this topic is crucial as it forms the bedrock for explaining how energy transfer affects substances, leading to either a temperature change or a change of state. Examiners frequently test this through multi-step calculations and graph interpretation, so a solid grasp is essential for success. This guide will break down the two key energy stores, explain the mathematical relationships you need, and show you how to apply them to exam-style questions, connecting directly to the assessment objectives (AO1, AO2, and AO3) to maximise your marks.
Key Concepts
Concept 1: Internal Energy as Kinetic + Potential Energy
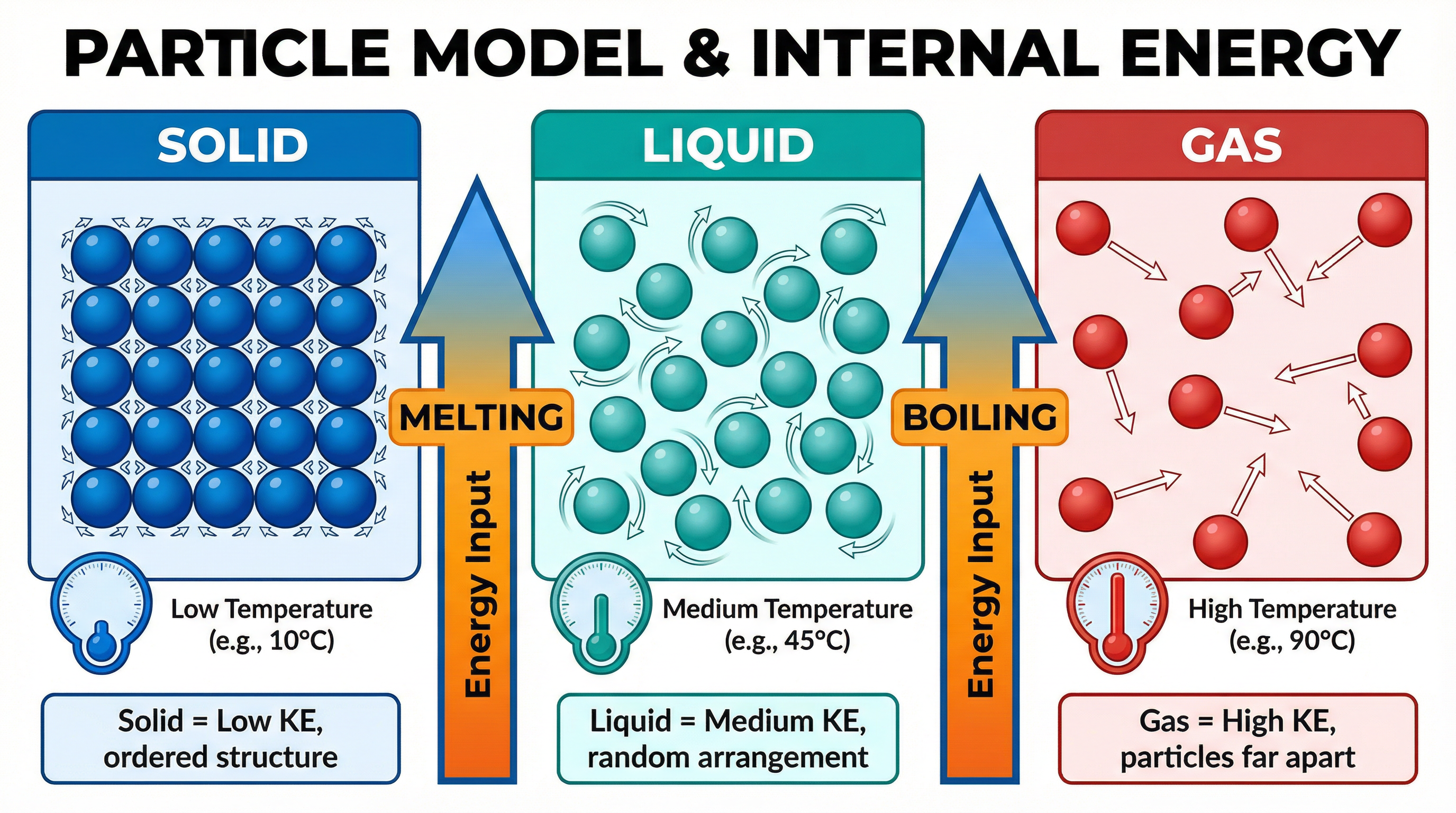
At the heart of this topic is the core definition. Internal energy isn’t just about how hot something is; it’s the sum of two distinct energy stores within the particles of a substance.
-
Kinetic Energy Store: This is the energy particles have due to their random motion. In solids, this is vibrational energy. In liquids and gases, it includes translational (moving from place to place) and rotational energy. Temperature is a measure of the average kinetic energy of the particles. When you heat a substance and its temperature rises, you are increasing the kinetic energy store of its particles.
-
Potential Energy Store: This is the energy stored due to the forces between particles (intermolecular forces). It relates to the particles' positions relative to each other. To change a substance's state (e.g., melting a solid to a liquid), you must overcome these forces and move the particles further apart. This requires energy, which is transferred to the potential energy store.
Concept 2: Changes of State and Latent Heat
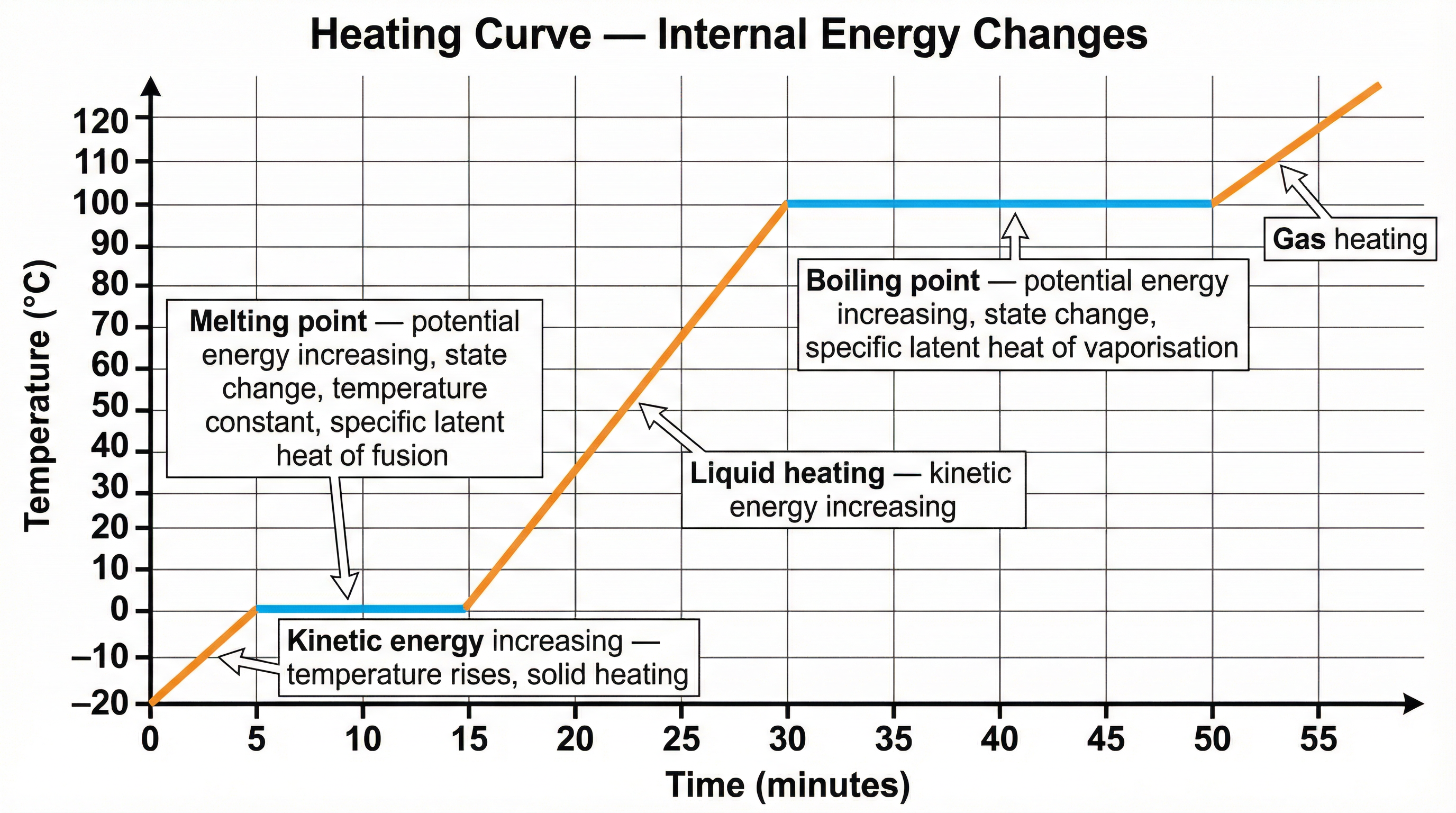
A change of state is a physical process where a substance changes from one state (solid, liquid, gas) to another. During a change of state, the temperature of the substance does not change. This is a critical point that candidates often misunderstand.
When a solid melts or a liquid boils, the energy being supplied is used to break the intermolecular bonds holding the particles together, not to increase their speed. This energy increases the potential energy store of the particles. The energy required for this process is called latent heat. 'Latent' means hidden, because the energy transfer doesn't cause a visible temperature change.
- Specific Latent Heat of Fusion (Lf): The energy required to change 1kg of a substance from solid to liquid with no change in temperature.
- Specific Latent Heat of Vaporisation (Lv): The energy required to change 1kg of a substance from liquid to gas with no change in temperature.
Mathematical/Scientific Relationships
Two key equations govern this topic. You must know when and how to use each one.
-
Specific Heat Capacity (for temperature changes)
- Formula:
ΔE = m × c × Δθ - Given on formula sheet?: Yes
- What it means:
ΔE(or Q) = Change in thermal energy (Joules, J)m= mass (kilograms, kg) — Examiner Tip: Watch out for mass given in grams! You MUST convert it.c= specific heat capacity (Joules per kilogram per degree Celsius, J/kg°C)Δθ= temperature change (degrees Celsius, °C)
- When to use it: Use this equation when a substance is being heated or cooled and its temperature is changing. Do NOT use it for a change of state.
- Formula:
-
Specific Latent Heat (for changes of state)
- Formula:
E = m × L - Given on formula sheet?: Yes
- What it means:
E(or Q) = Energy for a change of state (Joules, J)m= mass (kilograms, kg)L= specific latent heat (Joules per kilogram, J/kg). This will be eitherLffor fusion orLvfor vaporisation.
- When to use it: Use this equation ONLY when a substance is melting, freezing, boiling, or condensing at a constant temperature.
- Formula:
Practical Applications
This topic has many real-world applications, from engineering to everyday life.
-
Cooling Systems: Refrigerators and air conditioners work by exploiting changes of state. A special fluid (refrigerant) is vaporised (boiled) inside the unit. This process absorbs a large amount of thermal energy from the inside of the fridge (due to the high specific latent heat of vaporisation), making it cold. The gas is then pumped outside, compressed, and condensed back into a liquid, releasing the absorbed heat into the surroundings.
-
Climate Regulation: Large bodies of water, like oceans, have a very high specific heat capacity. This means they can absorb a huge amount of energy from the sun without a large temperature increase. They act as a thermal buffer, keeping coastal areas cooler in summer and warmer in winter than inland areas.
-
Required Practical: Investigating Specific Heat Capacity
- Apparatus: 1kg block of material (e.g., aluminium), thermometer, heater, ammeter, voltmeter, power pack, stopwatch, insulation.
- Method: Measure the mass of the block. Insert the heater and thermometer into the holes in the block. Wrap the block in insulation to reduce heat loss to the surroundings. Measure the initial temperature. Connect the heater to the power pack with the ammeter in series and voltmeter in parallel. Turn on the power and start the stopwatch. Record the voltage and current. After a set time (e.g., 10 minutes), turn off the power and record the final temperature. Calculate the energy supplied by the heater using
E = P × t = V × I × t. Calculate the temperature changeΔθ. Calculate the specific heat capacity usingc = ΔE / (m × Δθ). - Common Errors: Heat loss to the surroundings is the biggest source of error, leading to a calculated value of
cthat is higher than the true value. Not waiting for the temperature to distribute evenly through the block before taking the final reading is another error.