Study Notes

Overview
Latent heat sits at the heart of OCR GCSE Physics Topic 6.5 and represents one of the most conceptually rich areas of the thermal physics unit. The word 'latent' derives from the Latin latere, meaning 'to lie hidden' — and that etymology captures the central puzzle perfectly: energy is being transferred into or out of a substance, yet the thermometer reading refuses to budge. For candidates who understand the particle model, this is not mysterious at all; it is a precise and elegant consequence of how intermolecular forces work.
This topic connects directly to the particle model of matter, internal energy, and specific heat capacity — all of which appear elsewhere in the OCR specification. Examiners frequently set synoptic questions that require candidates to distinguish between latent heat and specific heat capacity, or to interpret a full heating curve that spans both concepts. The assessment objective breakdown for this topic is AO1 (30%), AO2 (40%), and AO3 (30%), meaning that application and analysis questions dominate — candidates must be able to use knowledge, not merely recall it.
Typical exam question styles include: (1) describe and explain a labelled section of a heating curve (2–4 marks); (2) calculate energy using E = mL with a unit conversion trap (3–4 marks); (3) explain in terms of particles why temperature is constant during a state change (3 marks); and (4) required practical questions about measuring specific latent heat using electrical methods (4–6 marks). Candidates who can answer all four styles fluently are well-positioned to achieve top marks.
Key Concepts
Concept 1: What is Latent Heat?
Latent heat is the energy transferred to or from a substance during a change of state, at constant temperature. It is 'latent' — hidden — because it does not manifest as a measurable temperature change. When a solid melts, energy is absorbed from the surroundings; when a liquid freezes, energy is released to the surroundings. In both cases, the temperature of the substance remains constant throughout the entire state change.
There are two distinct types of specific latent heat that candidates must know:
- Specific Latent Heat of Fusion (L_f): The energy required to change 1 kg of a substance from solid to liquid (or released when 1 kg changes from liquid to solid) at constant temperature. The term fusion refers to the solid–liquid transition. For water, L_f = 334,000 J/kg.
- Specific Latent Heat of Vaporisation (L_v): The energy required to change 1 kg of a substance from liquid to gas (or released when 1 kg changes from gas to liquid) at constant temperature. The term vaporisation refers to the liquid–gas transition. For water, L_v = 2,260,000 J/kg.
Note that the specific latent heat of vaporisation for water is approximately 6.8 times larger than the specific latent heat of fusion. This is because vaporisation requires completely overcoming all intermolecular forces so that particles can escape the liquid entirely, whereas fusion only partially weakens those forces.
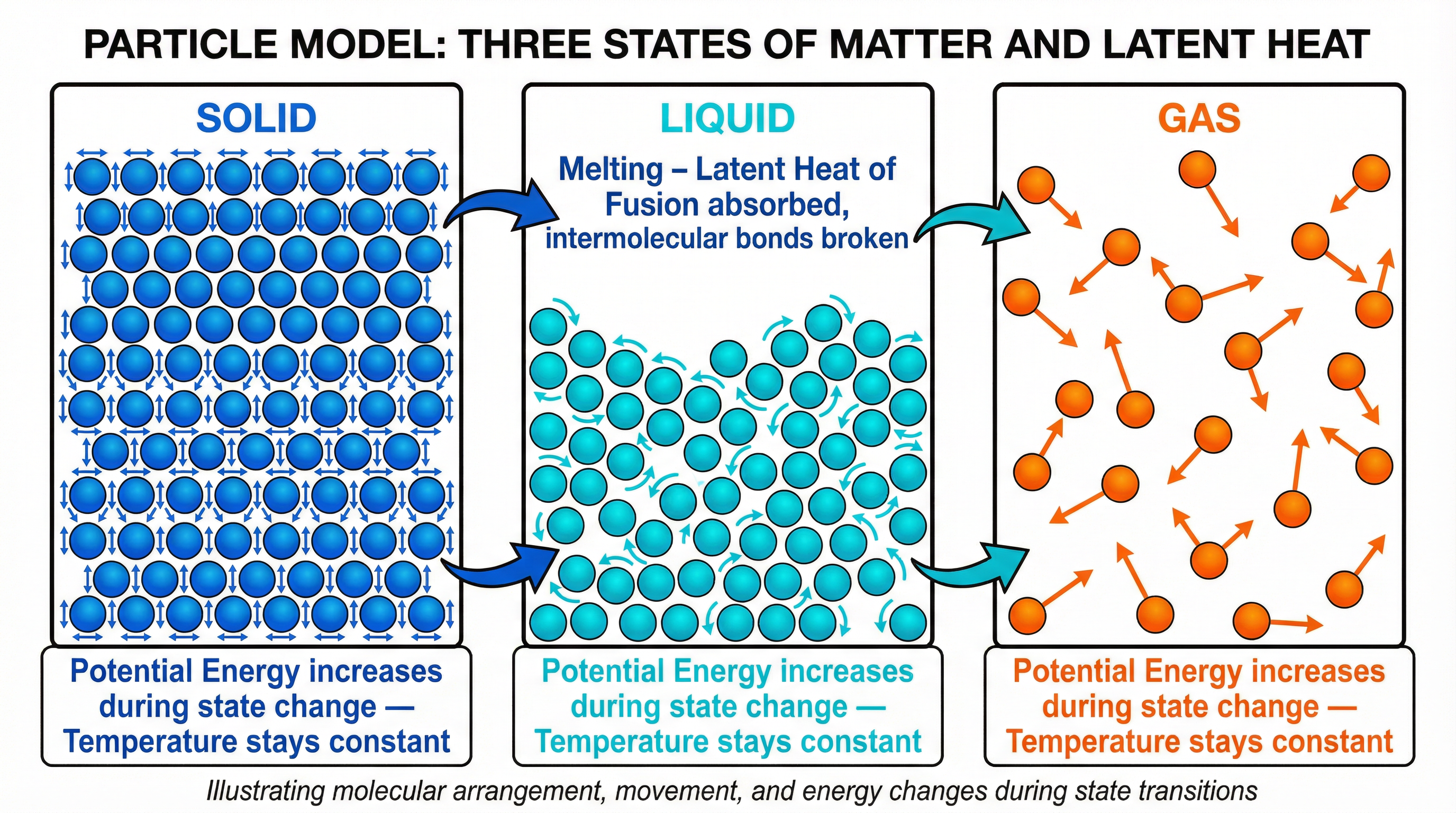
Concept 2: The Particle Model Explanation
The particle model provides the essential mechanistic explanation that earns marks in 'explain' questions. In a solid, particles are held in fixed positions by strong intermolecular forces (forces of attraction between particles). They vibrate about fixed points but cannot move freely. The temperature of a substance is a measure of the average kinetic energy of its particles — higher temperature means faster-moving particles.
When a solid is heated to its melting point and energy continues to be supplied, that energy is used to break the intermolecular bonds — to work against the forces of attraction and pull particles further apart. This process increases the potential energy of the particles (they are being separated against an attractive force, analogous to stretching a spring). Crucially, the kinetic energy of the particles does not increase during this process — the particles are not moving faster. Since temperature is determined by kinetic energy, the temperature remains constant.
Once all intermolecular bonds have been sufficiently weakened and the substance has fully changed state, further energy input once again increases kinetic energy, and the temperature rises.
The examiner's key distinction: During a state change, internal energy increases because potential energy increases, while kinetic energy remains constant. Temperature does not change because temperature reflects kinetic energy only.
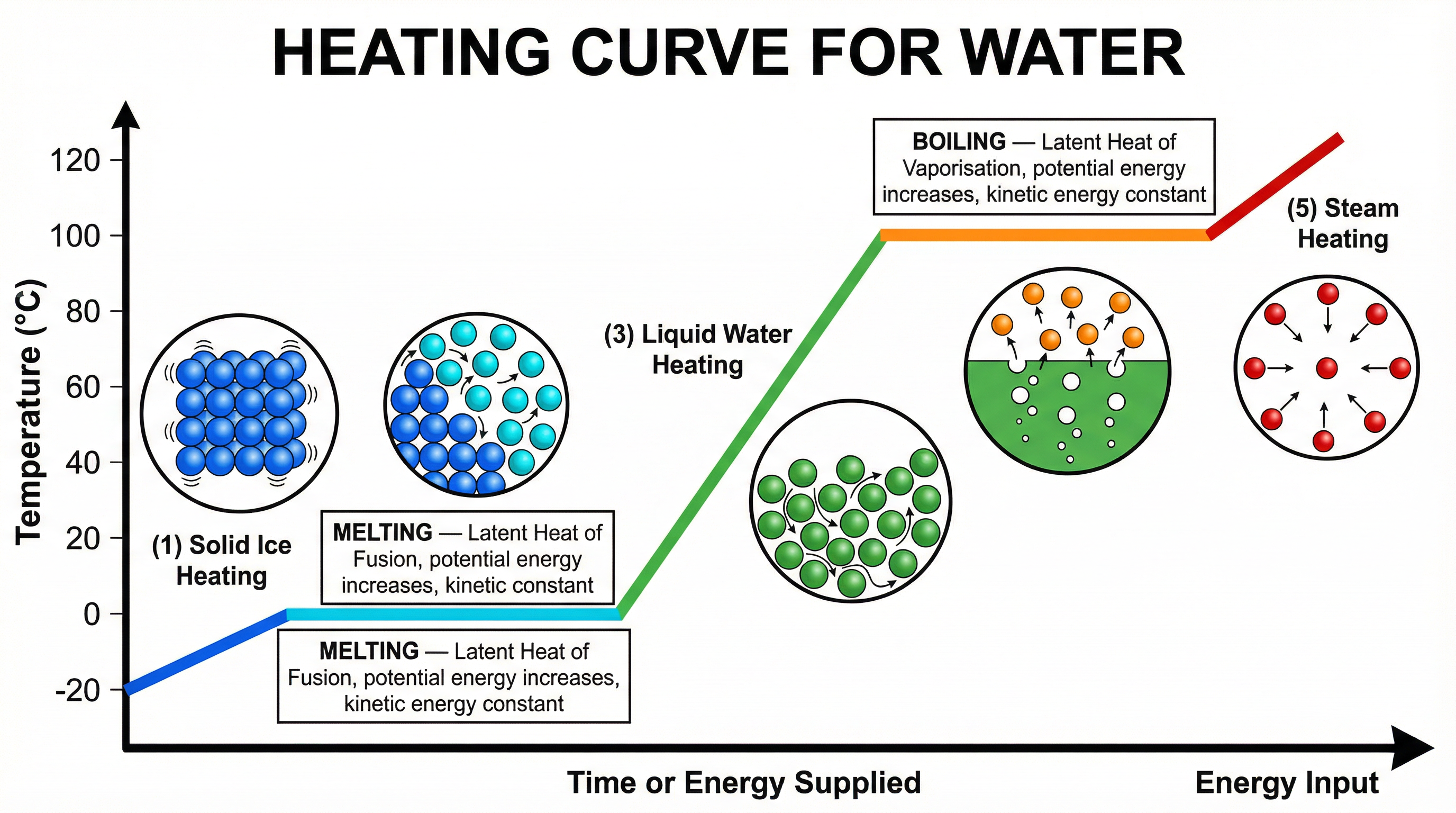
Concept 3: Heating and Cooling Curves
A heating curve plots temperature (y-axis) against time or energy supplied (x-axis) as a substance is heated from below its melting point to above its boiling point. For water, the five distinct sections are:
| Section | Temperature Range | Process | Energy Change |
|---|---|---|---|
| 1 | −20°C → 0°C | Ice heating (solid) | KE increases, temperature rises |
| 2 | 0°C (flat) | Melting | PE increases, KE constant, temperature constant |
| 3 | 0°C → 100°C | Water heating (liquid) | KE increases, temperature rises |
| 4 | 100°C (flat) | Boiling | PE increases, KE constant, temperature constant |
| 5 | >100°C | Steam heating (gas) | KE increases, temperature rises |
The horizontal (flat) sections are the latent heat regions. The length of the plateau at 100°C is longer than at 0°C because the specific latent heat of vaporisation is much greater than that of fusion — more energy must be supplied before all the liquid has converted to gas.
A cooling curve is the reverse: a substance cools from gas to solid. The flat sections appear at the same temperatures (100°C and 0°C for water) but now represent energy being released to the surroundings as bonds form.
Concept 4: The Formula E = mL
The quantitative relationship for latent heat is:
E = mLWhere:
- E = energy transferred (J)
- m = mass of substance (kg) — must be in kilograms when L is in J/kg
- L = specific latent heat (J/kg)
This formula can be rearranged to find any unknown:
- To find L: L = E ÷ m
- To find m: m = E ÷ L
Critical unit conversion: If mass is given in grams, divide by 1000 to convert to kilograms. This is the most frequently penalised error in OCR mark schemes. Always write the conversion as an explicit step in your working.
Formula status: E = mL must be memorised — it is not provided on the OCR formula sheet.
Mathematical/Scientific Relationships
| Formula | Variables | When to Use | Status |
|---|---|---|---|
| E = mL | E (J), m (kg), L (J/kg) | Change of state (constant temperature) | Must memorise |
| E = mcΔT | E (J), m (kg), c (J/kg°C), ΔT (°C) | Temperature change (no state change) | Given on formula sheet |
Unit Conversions Candidates Commonly Get Wrong:
- Grams to kilograms: divide by 1000 (e.g., 500 g = 0.5 kg)
- Kilojoules to joules: multiply by 1000 (e.g., 167 kJ = 167,000 J)
- If L is given in J/g (rare), keep mass in grams — do NOT convert
Practical Applications
Required Practical: Measuring Specific Latent Heat of Fusion of IceThis required practical is a favourite for OCR exam questions. Candidates must know the method, expected results, and sources of error.
Apparatus: Crushed ice, beaker, immersion heater (or heating coil), joulemeter (or ammeter and voltmeter and stopwatch), balance, thermometer.
Method: (1) Weigh a known mass of crushed ice in a beaker. (2) Record the initial mass. (3) Connect the immersion heater and record the energy supplied using a joulemeter, or calculate E = IVt using ammeter, voltmeter, and stopwatch. (4) Allow the ice to melt completely. (5) Record the mass of water remaining. (6) Calculate the mass of ice melted (m = final mass − mass of water that was already liquid). (7) Calculate L = E ÷ m.
Expected result: L_f ≈ 334,000 J/kg for water.
Common errors: Not accounting for ice that melted before the heater was switched on (control experiment needed); heat losses to surroundings causing an overestimate of E; not ensuring all ice has melted before taking the final mass reading.
How examiners test it: Questions may ask candidates to identify a source of error and explain how it affects the calculated value of L, or to suggest an improvement to the method.
Real-World Applications of Latent Heat:
- Sweating: evaporation of sweat from skin absorbs latent heat of vaporisation, cooling the body
- Refrigerators: refrigerant fluid evaporates inside the fridge (absorbing latent heat, cooling the interior) and condenses outside (releasing latent heat)
- Ice packs in sports medicine: melting ice absorbs large amounts of energy from an injury site
- Steam burns: steam at 100°C releases latent heat of vaporisation when it condenses on skin, causing more severe burns than liquid water at 100°C