Study Notes
Overview
Welcome to your deep dive into Melting and Boiling, a fundamental topic in thermal physics. This area of the specification, 6.6, is crucial for both Foundation and Higher tier candidates as it explores the transitions between the three states of matter: solid, liquid, and gas. Understanding what happens to particles and energy during these changes is key to unlocking a significant number of marks. Examiners frequently test this through heating curve interpretation, particle model descriptions, and calculations using specific latent heat. This guide will equip you with the precise language, calculation strategies, and exam techniques required by OCR. We will also explore synoptic links to topics like Energy (Topic 3) and Particle Model of Matter (Topic 6.1), showing how concepts interconnect across the physics course. Get ready to master the difference between kinetic and potential energy stores and apply the E = mL equation with confidence.
Key Concepts
Concept 1: The Particle Model and States of Matter
The particle model is the foundation for understanding this topic. Examiners expect you to describe the arrangement and motion of particles in solids, liquids, and gases with specific terminology.
- Solids: Particles are arranged in a regular lattice structure. They are held in fixed positions by strong intermolecular forces of attraction. Their movement is restricted to vibrating about these fixed positions. They have the least amount of internal energy.
- Liquids: Particles are still close together but are randomly arranged. The intermolecular forces are weaker than in solids, allowing particles to move past one another. This is why liquids can flow and take the shape of their container.
- Gases: Particles are far apart and move rapidly and randomly. The intermolecular forces are negligible. They have the most internal energy and will fill any container they are in.
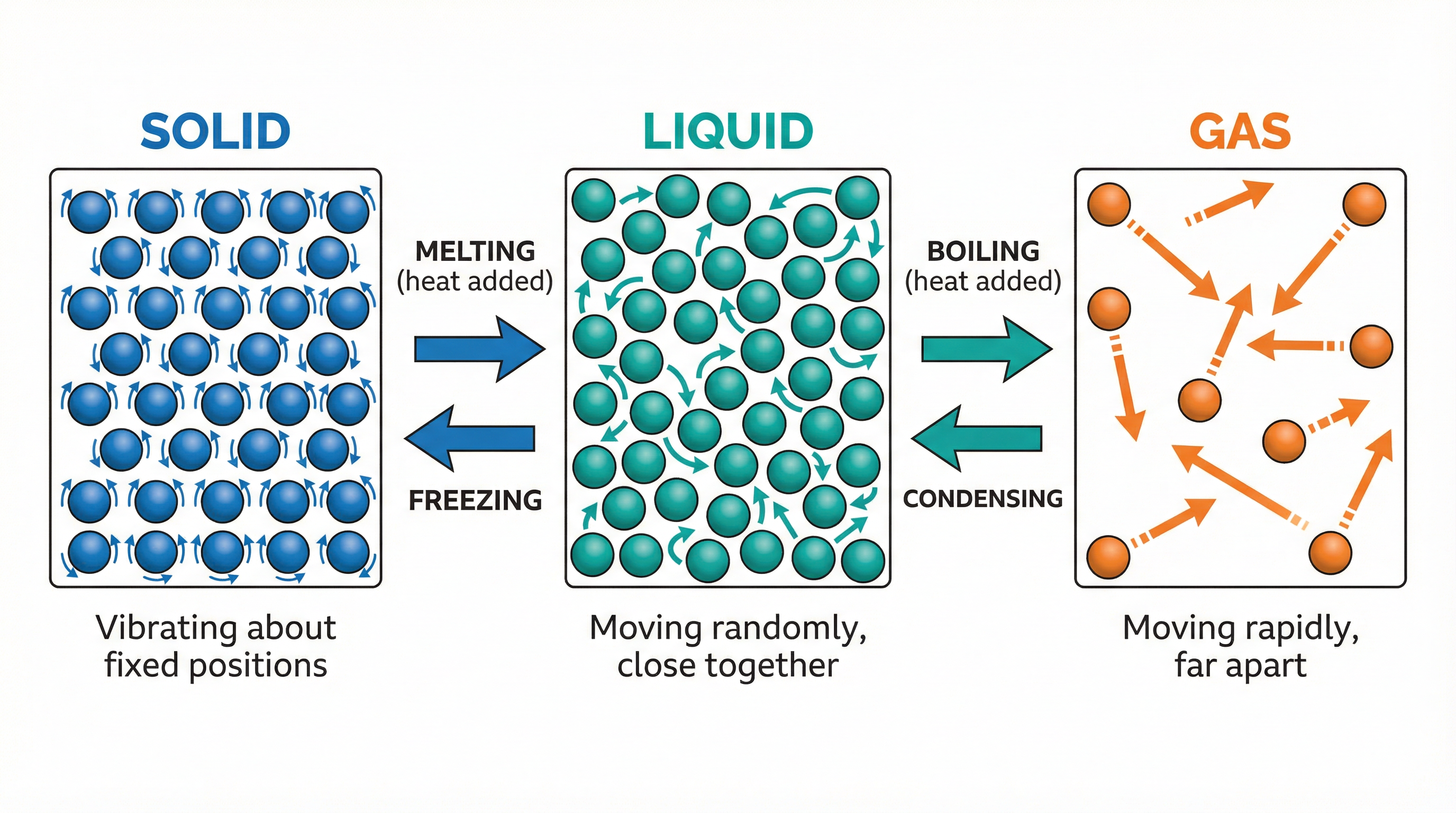
Why this matters: Questions worth 2-3 marks often ask you to 'Describe the differences in the arrangement and motion of particles...'. Using the bolded keywords is essential for credit.
Concept 2: Internal Energy (Kinetic vs. Potential)
Internal energy is the total energy stored by the particles of a substance. It has two components:
- Kinetic Energy: The energy an object has due to its motion. In this context, it relates to how fast the particles are moving or vibrating. Temperature is a measure of the average kinetic energy of the particles. When you heat a substance and its temperature rises, you are increasing the kinetic energy of its particles.
- Potential Energy: The energy stored due to the position of the particles relative to each other. It is associated with the intermolecular forces between particles. When you change the state of a substance (e.g., melting), you are changing the spacing and arrangement of particles, which alters their potential energy.
The Crucial Distinction: During a temperature change, kinetic energy changes. During a state change, potential energy changes while kinetic energy (and therefore temperature) remains constant.
Concept 3: Heating Curves and Changes of State
A heating curve is a graph of temperature against time for a substance as it is heated at a constant rate. It provides a visual representation of energy changes. Examiners love these graphs.
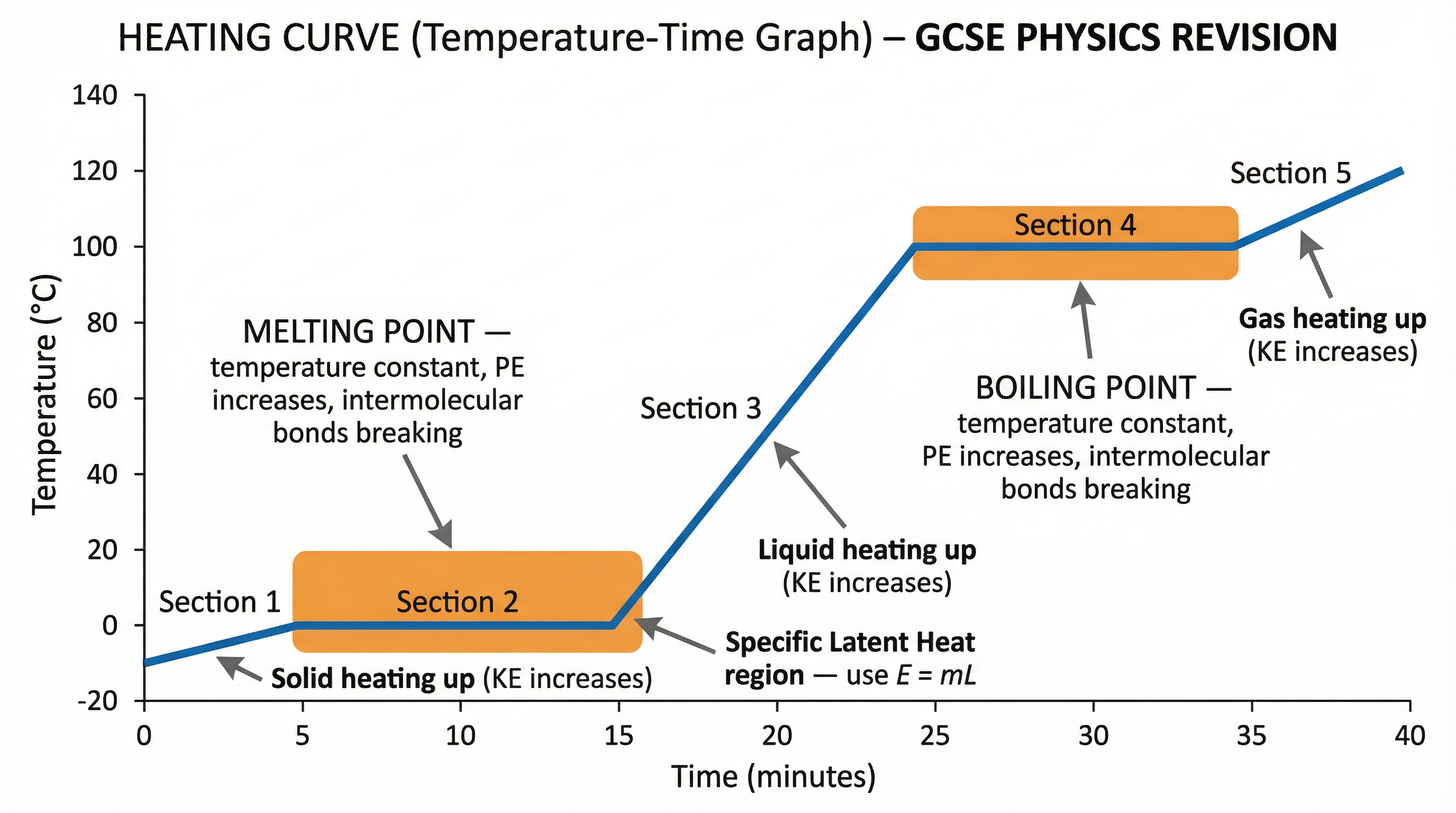
Let's break down the sections of the heating curve for water:
- Section 1 (Sloping Up): The solid (ice) is being heated. Energy input increases the kinetic energy of the particles, so they vibrate more vigorously. The temperature rises.
- Section 2 (Flat Plateau): The substance is at its melting point (0°C for water). The energy being supplied is now used to break the intermolecular bonds holding the particles in their fixed lattice. This increases the potential energy store, not the kinetic energy. Therefore, the temperature remains constant. This is a critical point that candidates often miss.
- Section 3 (Sloping Up): The substance is now a liquid (water). All the ice has melted. Further energy input increases the kinetic energy of the particles, so they move faster. The temperature rises again.
- Section 4 (Flat Plateau): The substance is at its boiling point (100°C for water). The energy supplied is used to overcome the remaining intermolecular forces and separate the particles completely as they turn into a gas (steam). This again increases the potential energy store. The temperature remains constant.
- Section 5 (Sloping Up): The substance is now a gas (steam). Further energy input increases the kinetic energy of the gas particles, so they move even faster. The temperature rises.
Mathematical/Scientific Relationships
There are two key equations for thermal energy calculations. Knowing which one to use is vital.
-
Specific Heat Capacity (When Temperature Changes)
Formula:E = m c Δθ
Status: Given on formula sheet
When to use: For any sloping section of a heating/cooling curve where the temperature is changing.
Symbol Meanings:
-E: Energy transferred (Joules, J)
-m: Mass of the substance (kilograms, kg)
-c: Specific Heat Capacity (Joules per kilogram per degree Celsius, J/kg°C)
-Δθ: Change in temperature (degrees Celsius, °C) -
Specific Latent Heat (When State Changes)
Formula:E = m L
Status: Given on formula sheet
When to use: For any flat section of a heating/cooling curve where the state is changing (melting, boiling, freezing, condensing).
Symbol Meanings:
-E: Energy transferred (Joules, J)
-m: Mass of the substance (kilograms, kg)
-L: Specific Latent Heat (Joules per kilogram, J/kg)
There are two types of Specific Latent Heat:
- Specific Latent Heat of Fusion (L_f): The energy needed to change 1kg of a substance from solid to liquid without a change in temperature.
- Specific Latent Heat of Vaporisation (L_v): The energy needed to change 1kg of a substance from liquid to gas without a change in temperature.
Practical Applications
This topic is not just theoretical; it explains many real-world phenomena.
- Sweating: When you exercise, your body produces sweat. As the sweat (liquid) evaporates from your skin, it turns into a gas. This process requires energy, which it takes from your body. This is an application of latent heat of vaporisation, and it's why sweating cools you down.
- Refrigeration: Refrigerators work by circulating a special fluid. Inside the fridge, this fluid evaporates (boils at a low temperature), absorbing heat from the food compartment (latent heat of vaporisation). Outside the fridge, at the back, the gas is compressed and condenses back into a liquid, releasing this heat into the room.
- Cooking: When you boil water to cook pasta, the water temperature stays at 100°C no matter how vigorously it boils. Turning the heat up higher just makes it boil away faster (supplying the latent heat of vaporisation more quickly) but doesn't cook the pasta any quicker.
Required Practical: Although there isn't a single named required practical for this specific topic in OCR GCSE Physics, skills related to measuring temperature and energy changes are fundamental. A common classroom experiment involves measuring the specific latent heat of fusion of ice.
Apparatus: Calorimeter, thermometer, top-pan balance, ice, heater.
Method: Measure the mass of water in a calorimeter. Record the initial temperature. Add a known mass of ice and wait for it to melt. Record the final temperature. The energy lost by the water can be calculated (E=mcΔθ) and equated to the energy gained by the ice to melt (E=mL). This allows for a calculation of L. Common errors include heat loss to the surroundings and inaccurate temperature readings.