Study Notes
Overview
Radioactive decay is a fundamental concept in nuclear physics, describing how unstable atomic nuclei release energy to become more stable. For your OCR GCSE Physics exam, a thorough understanding of the three main types of decay—alpha (α), beta (β), and gamma (γ)—is essential. This topic is not just about memorising properties; it's about applying that knowledge to balance nuclear equations, understand safety precautions, and explain real-world applications. Examiners will test your ability to compare the ionising and penetrating power of each radiation type and link these properties to their uses, such as in smoke detectors and medical treatments. This guide will walk you through the core principles, provide examiner insights, and equip you with the skills to tackle any question on radioactive decay with confidence. Expect to see a mix of short-answer definition questions (AO1), application questions (AO2), and longer evaluation questions (AO3).
Key Concepts
Concept 1: The Unstable Nucleus and Random Decay
At the heart of radioactivity is the unstable nucleus. An atomic nucleus can be unstable if it has an imbalance of protons and neutrons, or simply too much energy. To achieve stability, it spontaneously emits radiation. A crucial point that candidates must remember is that this process is entirely random and spontaneous. You cannot predict which nucleus will decay next, nor can you influence the rate of decay by changing physical conditions like temperature, pressure, or by chemical reactions. This is a common misconception tested by examiners.
Examiner's Note: Credit is often given for explicitly stating that decay is a nuclear process, unaffected by external factors. A typical 1-mark question might be: "State two factors that do not affect the rate of radioactive decay."
Concept 2: Alpha (α) Decay
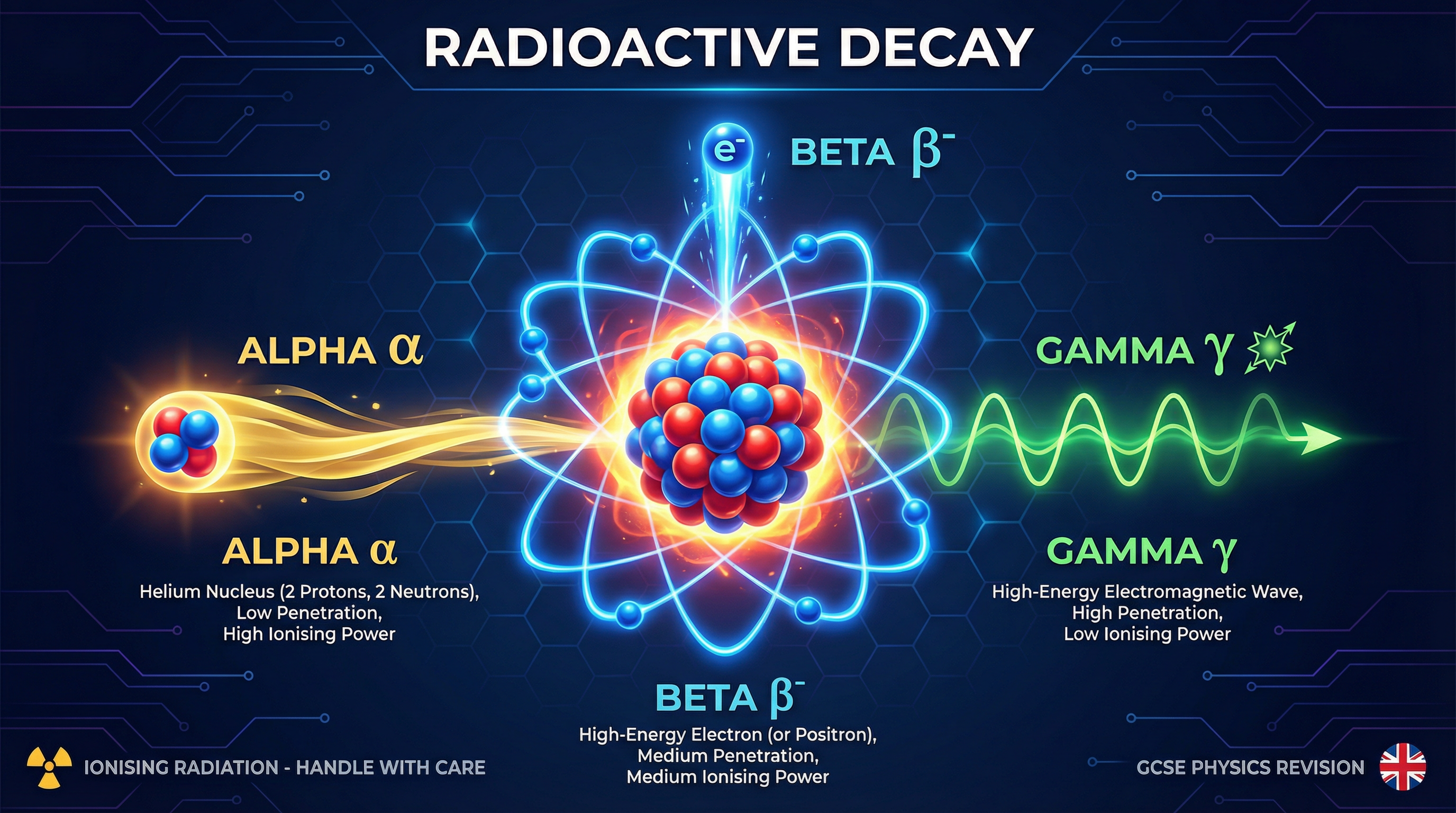
An alpha particle is a helium nucleus, composed of 2 protons and 2 neutrons. It has a relative mass of 4 and a charge of +2. When a nucleus undergoes alpha decay, it ejects an alpha particle.
- Effect on Nucleus: The mass number (top number) decreases by 4. The atomic number (bottom number) decreases by 2. This means the element itself changes, a process called transmutation.
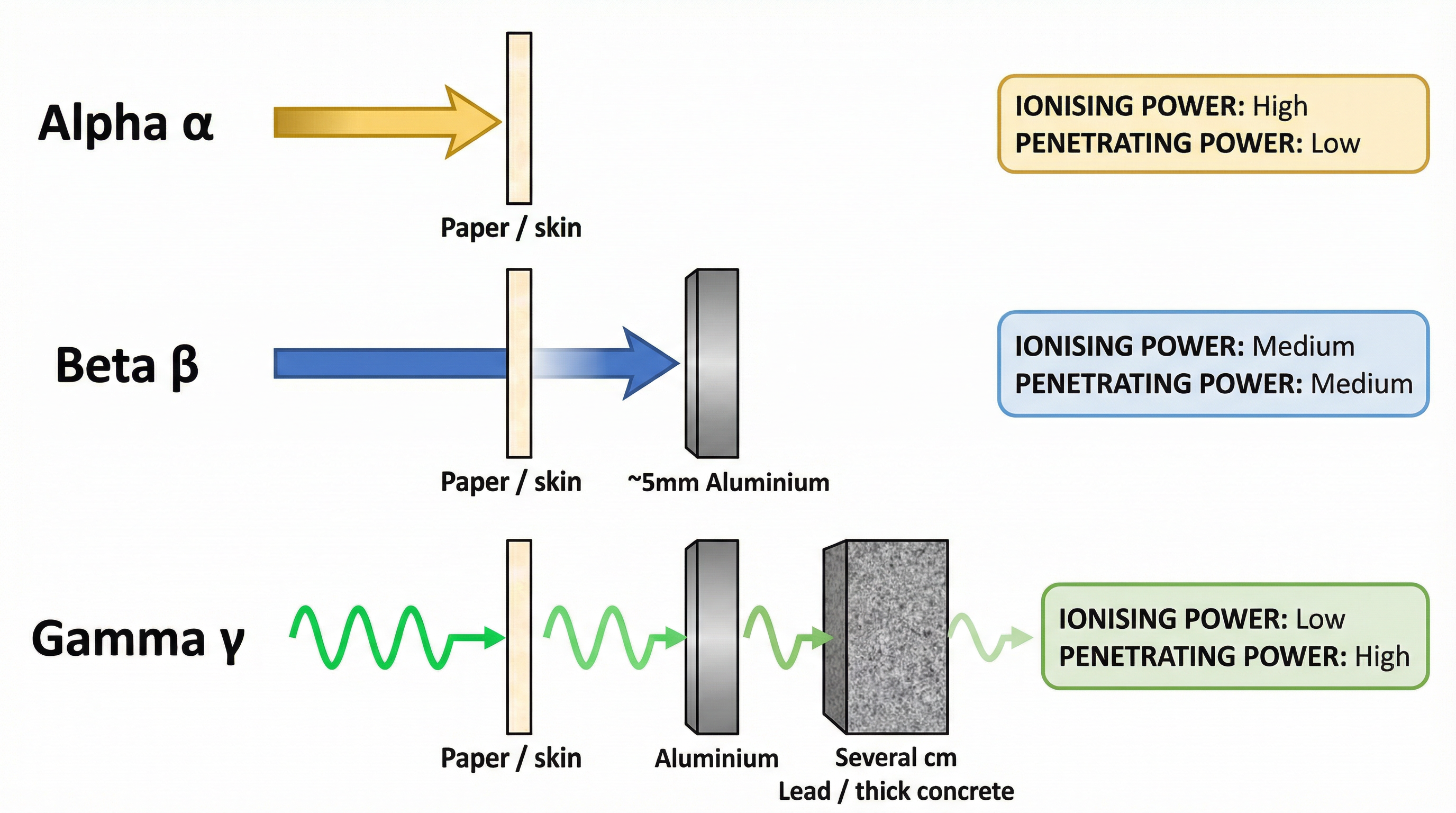
- Properties: Due to their size and +2 charge, alpha particles are highly ionising. They readily knock electrons off atoms they collide with. However, this causes them to lose energy quickly, giving them very low penetrating power. They are stopped by just a few centimetres of air or a single sheet of paper.
Example: Uranium-238 decays into Thorium-234.
Concept 3: Beta (β) Decay
A beta particle is a high-energy electron ejected from the nucleus. This is a critical point: it originates from the nucleus, not the electron shells. Beta decay occurs when a neutron in the nucleus turns into a proton.
- Effect on Nucleus: The mass number remains unchanged (as one neutron is lost but one proton is gained). The atomic number increases by 1 (due to the new proton).
- Properties: Beta particles are much smaller and faster than alpha particles. They have a charge of -1. They are moderately ionising and have moderate penetrating power. They can travel several metres in air and are stopped by about 5mm of aluminium.
Example: Carbon-14 decays into Nitrogen-14.
Concept 4: Gamma (γ) Emission
Gamma radiation is not a particle; it is a high-energy electromagnetic wave. It has no mass and no charge. Gamma emission often occurs after an alpha or beta decay event, as it is a way for the newly formed nucleus to release excess energy and settle into a more stable state.
- Effect on Nucleus: There is no change to the mass number or the atomic number of the nucleus.
- Properties: Because they have no charge or mass, gamma rays are weakly ionising. They do not interact strongly with atoms. This same property gives them very high penetrating power. They can only be significantly reduced by several centimetres of lead or metres of concrete.
Mathematical/Scientific Relationships
Balancing nuclear equations is a core mathematical skill for this topic. The fundamental rule is the conservation of mass number and atomic number.
The Golden Rule: The sum of the mass numbers (top) on the left side of the equation must equal the sum on the right. The sum of the atomic numbers (bottom) on the left must also equal the sum on the right.

Formulas (Must Memorise)
-
Alpha Decay General Form:
- A/Z X → (A-4)/(Z-2) Y + 4/2 α
- Where X is the parent nucleus and Y is the daughter nucleus.
-
Beta Decay General Form:
- A/Z X → A/(Z+1) Y + 0/-1 β (or 0/-1 e)
- Where X is the parent nucleus and Y is the daughter nucleus.
-
Gamma Emission General Form:
- A/Z X* → A/Z X + γ
- The asterisk (*) denotes an excited nucleus.
Practical Applications
Examiners expect you to link the properties of each radiation type to its uses. Marks are awarded for justifying the choice of radiation.
| Application | Radiation Used | Property Justification | Tier |
|---|---|---|---|
| Smoke Detectors | Alpha (α) | High ionising power creates a current in the air. Low penetrating power means it is safely contained and won't escape the detector casing. | Both |
| Paper Thickness Gauge | Beta (β) | Moderate penetrating power allows it to pass through paper but be affected by thickness changes. Alpha would be stopped; gamma would pass through unaffected. | Both |
| Sterilising Equipment | Gamma (γ) | High penetrating power allows it to penetrate packaging to kill all bacteria. As it is not a particle, it does not make the equipment radioactive. | Both |
| Medical Tracers | Gamma (γ) | High penetrating power allows it to be detected outside the body. Low ionising power minimises tissue damage. A short half-life is also crucial. | Higher |
| Cancer Treatment | Gamma (γ) | High penetrating power allows it to reach deep-seated tumours. The beam can be focused to kill cancer cells while minimising damage to healthy tissue. | Higher |
Safety: Irradiation vs. Contamination
A common source of confusion is the difference between irradiation and contamination.
- Irradiation: This is when an object or person is exposed to radiation from an external source. The exposure stops when the source is removed. Shielding (e.g., lead apron) and distance are the main protective measures.
- Contamination: This is when radioactive atoms get onto or into an object or person. The person is now a source of radiation. This is far more dangerous, especially with alpha emitters, as the internal organs are directly exposed to highly ionising radiation. Protective clothing (gloves, lab coat) and careful handling are required to prevent contamination.