Study Notes
Overview
Welcome to your definitive guide for OCR GCSE Physics topic 6.1: States of Matter. This topic is a cornerstone of thermal physics, and a solid understanding here will pay dividends across the course. We will explore the particle model (also known as kinetic theory) to explain the distinct properties of solids, liquids, and gases. A key focus for examiners is your ability to describe the arrangement, motion, and energy of particles in each state with precision. You will also need to apply the principle of mass conservation to changes of state and link these transitions to the concept of internal energy. Expect to see a mix of short-answer definition questions, descriptive comparisons, and structured 6-mark questions requiring you to explain a change of state from start to finish. This guide will equip you with the exact terminology and structured approach needed to excel.
Key Concepts
Concept 1: The Particle Model
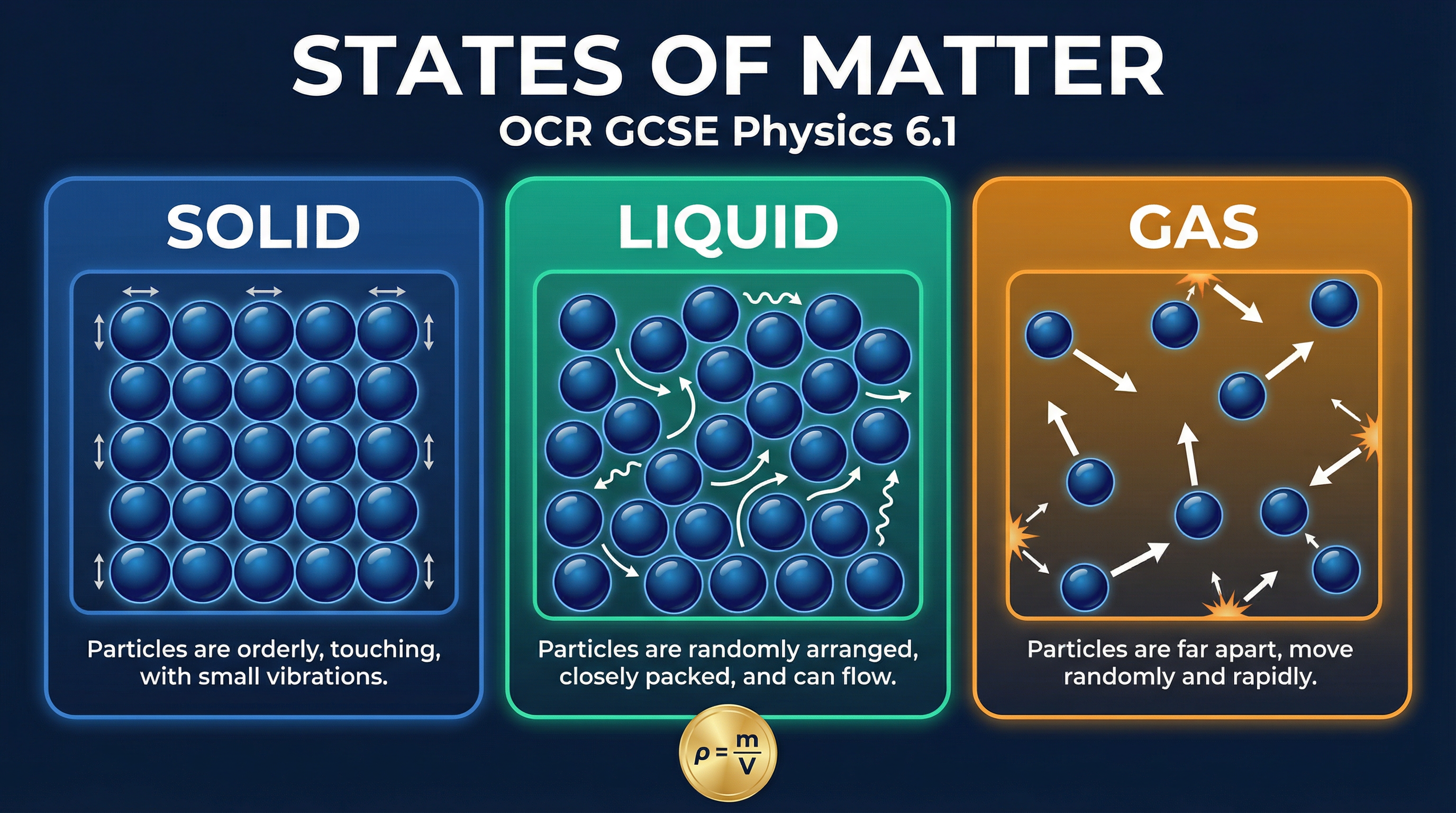
The particle model is the central theory you must use to explain the properties of matter. It states that all substances are made of a vast number of tiny, moving particles. The energy of these particles and the forces between them determine the state of the substance. Temperature is a measure of the average kinetic energy of the particles. The higher the temperature, the more kinetic energy the particles have, and the faster they move or vibrate.
-
Solids: Particles are arranged in a fixed, regular lattice. They are held in place by strong intermolecular forces, meaning they cannot move from their positions. However, they are not motionless. Crucially, they vibrate about their fixed positions. This fixed arrangement gives solids a definite shape and volume. Due to the tightly packed particles, solids have a high density.
-
Liquids: Particles are arranged irregularly and are in close contact with each other. The intermolecular forces are weaker than in solids, allowing the particles to move past one another. This is why liquids can flow and take the shape of their container. However, the forces are still strong enough to keep the particles together, giving liquids a fixed volume. When drawing diagrams, it is vital that candidates show liquid particles as being mostly in contact, as this explains why they are virtually incompressible. Their density is typically slightly lower than the solid state (water is a key exception).
-
Gases: Particles are arranged randomly and are very far apart from each other. The intermolecular forces are negligible (effectively zero). This means particles move rapidly and randomly in all directions, colliding with each other and the container walls. This movement is why gases fill any container they are in and have no fixed shape or volume. Due to the large spaces between particles, gases have a very low density and are easily compressed.
Concept 2: Changes of State & Internal Energy
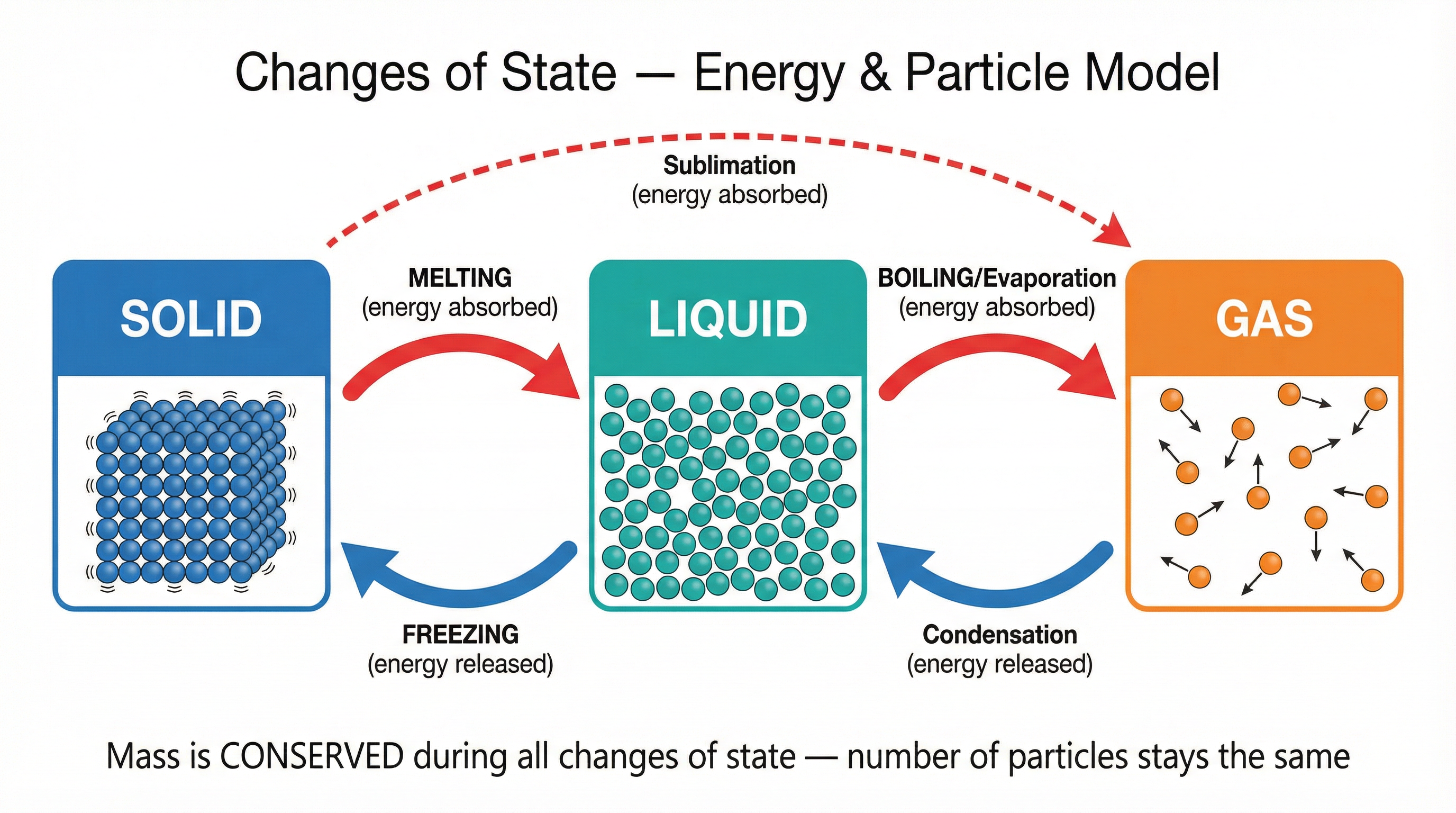
A change of state is a physical change, meaning no new substance is formed and the process is reversible. Mass is always conserved during these changes because the number of particles remains constant.
Changes of state involve energy transfer. The internal energy of a substance is the total energy stored in its particles — it is the sum of their kinetic energy (due to their motion) and their potential energy (due to the bonds between them).
-
Heating a Substance: When you heat a substance, you transfer energy to its internal energy store. This can either increase the particles' kinetic energy (raising the temperature) OR increase their potential energy (breaking bonds to change the state).
-
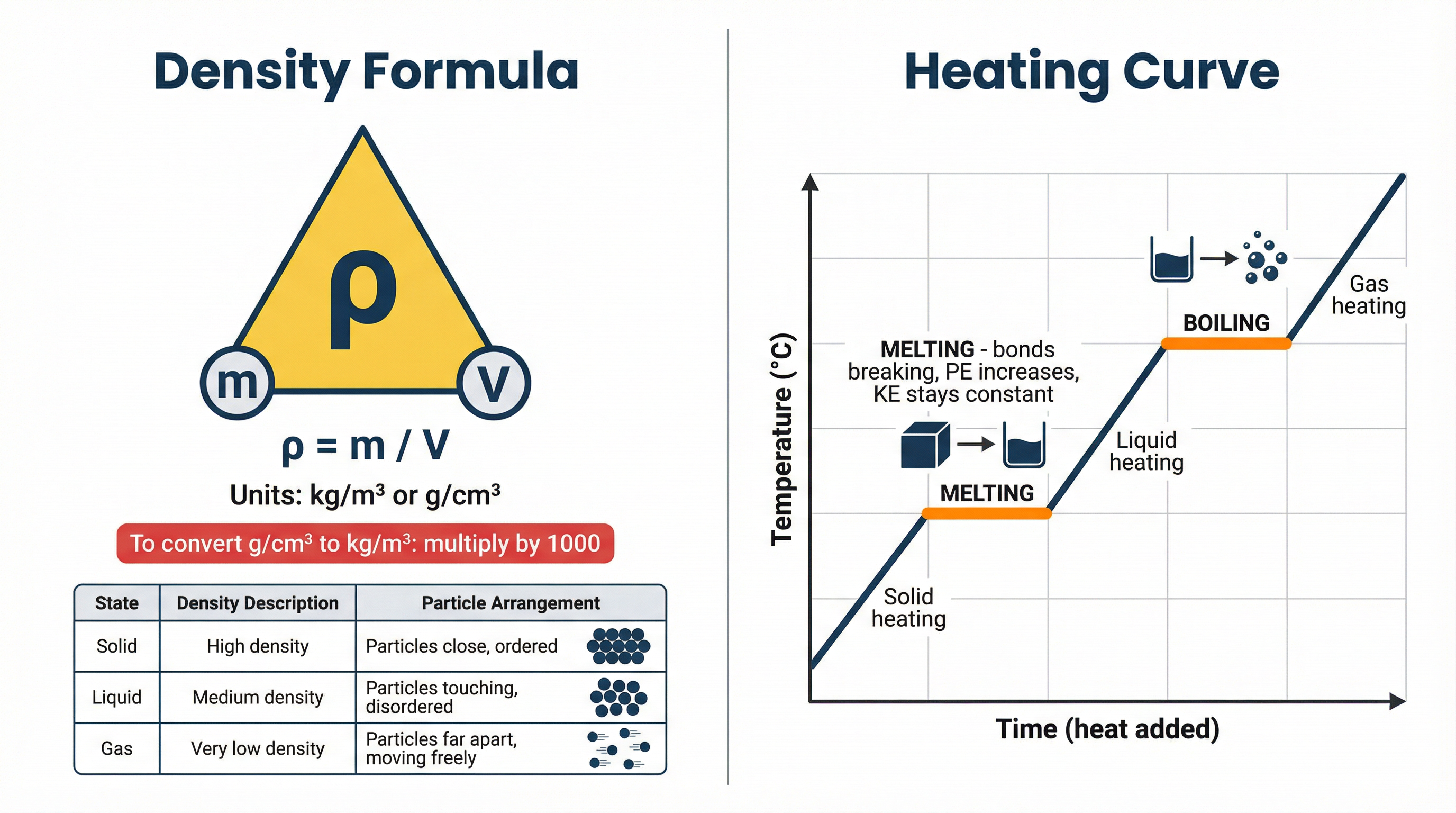
Melting & Boiling: During melting (solid to liquid) or boiling (liquid to gas), the energy supplied is used to overcome the intermolecular forces holding the particles together. This increases the potential energy of the particles, but not their kinetic energy. Therefore, during a change of state, the temperature of the substance does not change. This is represented by the flat sections on a heating curve.
-
Freezing & Condensing: During freezing (liquid to solid) or condensing (gas to liquid), bonds are formed between particles. This process releases energy, causing the potential energy of the particles to decrease. This energy release means the substance cools down unless the surrounding temperature is also at the freezing/boiling point.
Mathematical/Scientific Relationships
Density
Density is a measure of how much mass is packed into a certain volume. It is a key property that helps distinguish between the states of matter.
- Formula: Density (ρ) = Mass (m) / Volume (V)
- Symbol Meanings:
- ρ (rho) = Density, measured in kilograms per cubic metre (kg/m³) or grams per cubic centimetre (g/cm³).
- m = Mass, measured in kilograms (kg) or grams (g).
- V = Volume, measured in cubic metres (m³) or cubic centimetres (cm³).
- Status: This formula must be memorised. It is not always given on the formula sheet.
Unit Conversions
Examiners frequently test unit conversions for density. A common error is to miscalculate the conversion between g/cm³ and kg/m³.
- 1 g/cm³ = 1000 kg/m³To understand this, consider a 1m x 1m x 1m cube. Its volume is 1 m³. Since 1m = 100cm, its volume is also 100cm x 100cm x 100cm = 1,000,000 cm³. If a substance has a density of 1 g/cm³, then 1,000,000 cm³ of it has a mass of 1,000,000 g, which is 1000 kg. So, 1000 kg occupies 1 m³, meaning the density is 1000 kg/m³.
Practical Applications
Understanding states of matter is crucial for many real-world applications, from engineering to cooking. For example, the expansion and contraction of materials with temperature changes (due to changes in particle vibration and spacing) must be accounted for when designing bridges and railway lines. The principles of boiling and condensation are fundamental to power stations, refrigerators, and air conditioning systems.
Required Practical: Determining Density
OCR Gateway Physics includes a required practical to determine the density of regular and irregular objects.
- Apparatus: For a regular object (e.g., a cube): 30cm ruler, digital balance. For an irregular object (e.g., a rock): eureka can (displacement can), measuring cylinder, digital balance, water.
- Method (Regular Object):
- Measure the mass of the object using the digital balance. Record in kg.
- Use the ruler to measure the length, width, and height of the object in metres.
- Calculate the volume: Volume = length × width × height.
- Use the formula ρ = m/V to calculate the density.
- Method (Irregular Object):
- Measure the mass of the object using the digital balance.
- Fill the eureka can with water until water starts to trickle out of the spout. Wait for it to stop dripping.
- Place an empty measuring cylinder under the spout.
- Carefully lower the object into the eureka can. It will displace a volume of water equal to its own volume.
- Collect the displaced water in the measuring cylinder and read the volume. This is the volume of the object.
- Use the formula ρ = m/V to calculate the density.
- Common Errors: Parallax error when reading the ruler or measuring cylinder. Not waiting for the eureka can to stop dripping initially. Splashing water when immersing the object. Forgetting to convert units to be consistent (e.g., using mass in g and volume in cm³ is fine, but mixing g with m³ is not).