Study Notes

Overview
This study guide focuses on the core scientific principles that underpin all food preparation, a cornerstone of the OCR GCSE specification. Understanding the functional and chemical properties of ingredients is not just about knowing what happens, but why it happens. Examiners expect candidates to articulate these changes with scientific precision, moving beyond simple descriptions to detailed explanations. This guide will equip you with the language and concepts to explain how carbohydrates like starch behave (gelatinisation, dextrinisation) and how proteins in flour and eggs transform (gluten formation, coagulation). Mastering this topic is crucial for achieving high grades, as it demonstrates the deeper, analytical understanding required for top-level responses. We will break down these complex processes into clear, memorable steps, complete with the critical temperatures and terminology that will earn you credit.
Key Chemical Processes
Carbohydrates: Starch & Sugar
Gelatinisation: This is the process where starch thickens a liquid. It is fundamental to making sauces, custards, and gravies. Candidates must know the specific temperature points for full marks.
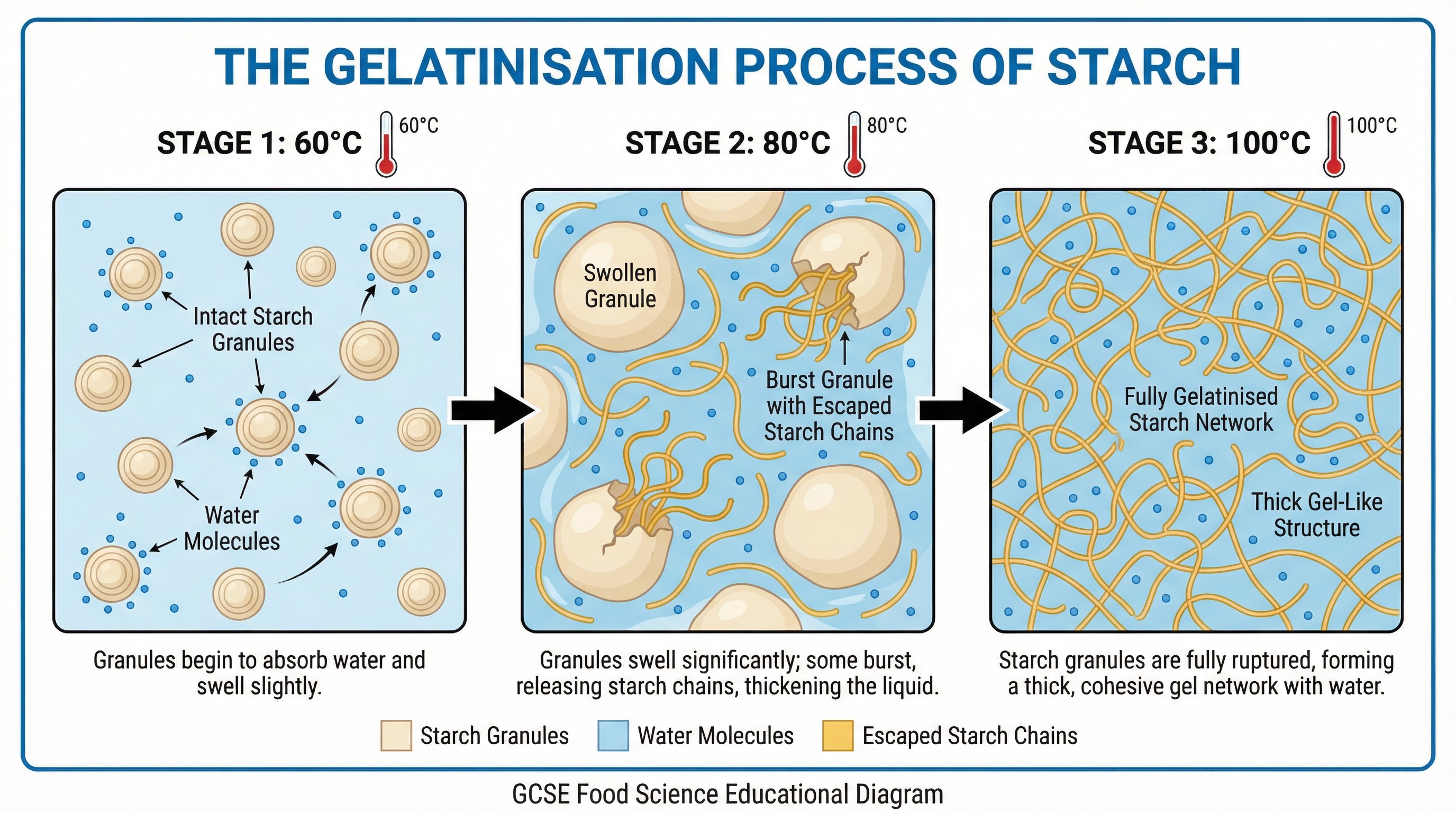
- Process: Starch granules are heated in a liquid.
- Reaction: At 60°C, granules start to absorb water and swell. At 80°C, they burst, releasing long starch molecules that tangle and trap water. The process is complete at 100°C, forming a thick, viscous gel.
- Outcome: A thickened, smooth sauce or gel.
Dextrinisation: This is the browning of starch with dry heat. It is responsible for the colour and flavour of toast and baked goods.
- Process: Starch is exposed to dry heat (e.g., grilling, baking).
- Reaction: The long starch molecules break down into smaller molecules called dextrins.
- Outcome: A golden-brown colour and a slightly sweet, nutty flavour.
Caramelisation: This is the browning of sugar with heat. It is distinct from dextrinisation.
- Process: Sugar is heated to a high temperature (around 160-180°C).
- Reaction: The sugar molecules break down and reform into new compounds.
- Outcome: A rich brown colour and complex caramel flavour.
Proteins: Gluten & Coagulation
Gluten Formation: Essential for bread-making, this process creates the structure needed for dough to rise.
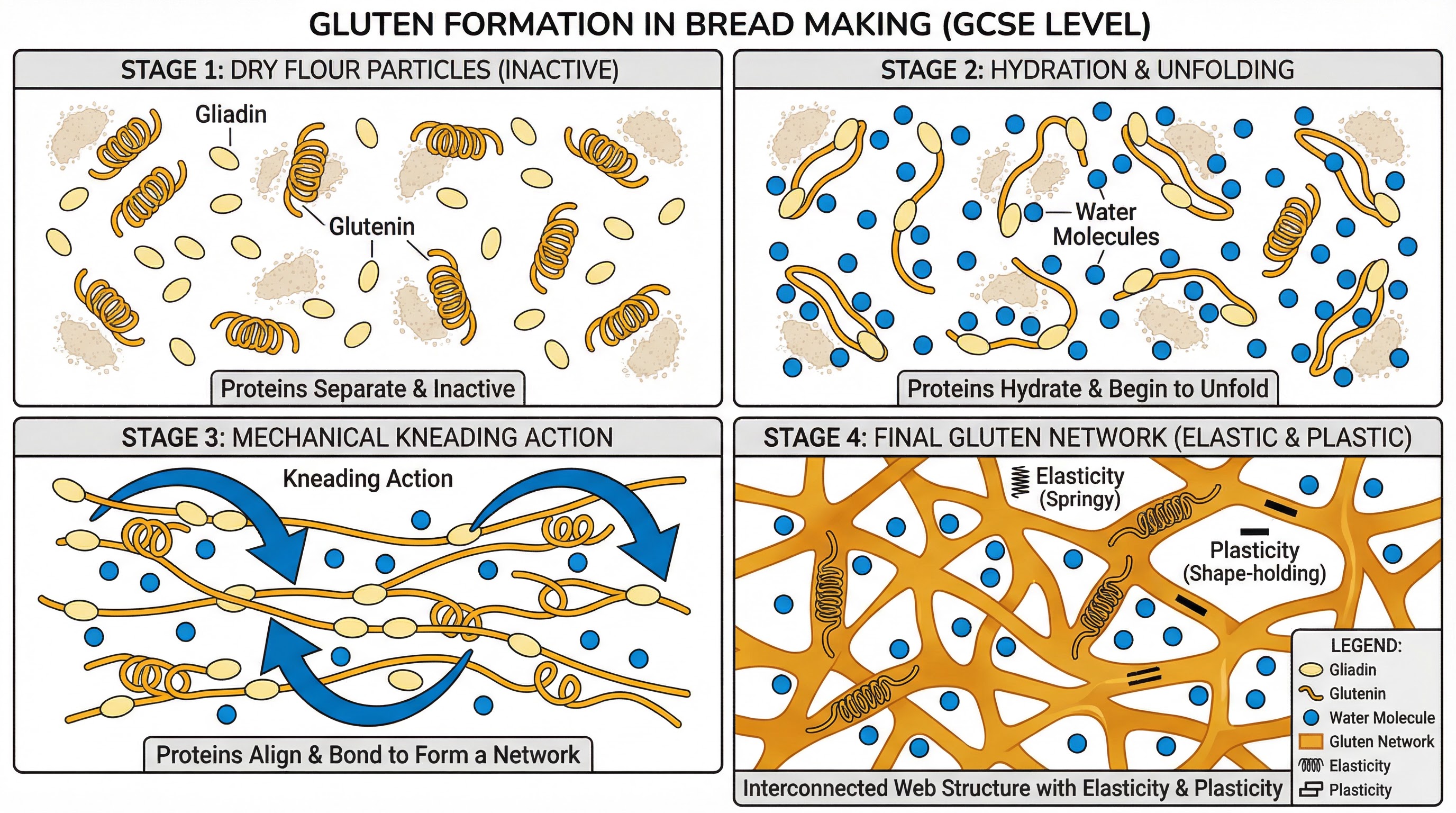
- Process: Wheat flour (containing proteins gliadin and glutenin) is mixed with water and kneaded.
- Reaction: Hydration allows the gliadin and glutenin proteins to unwind. Mechanical action (kneading) stretches and aligns these proteins, causing them to link together and form a viscoelastic network called gluten.
- Outcome: A dough that is both elastic (can spring back) and plastic (can be moulded), capable of trapping CO2 gas from yeast.
Coagulation: This is the setting or solidifying of proteins when heated. It is vital for cooking eggs, meat, and fish.
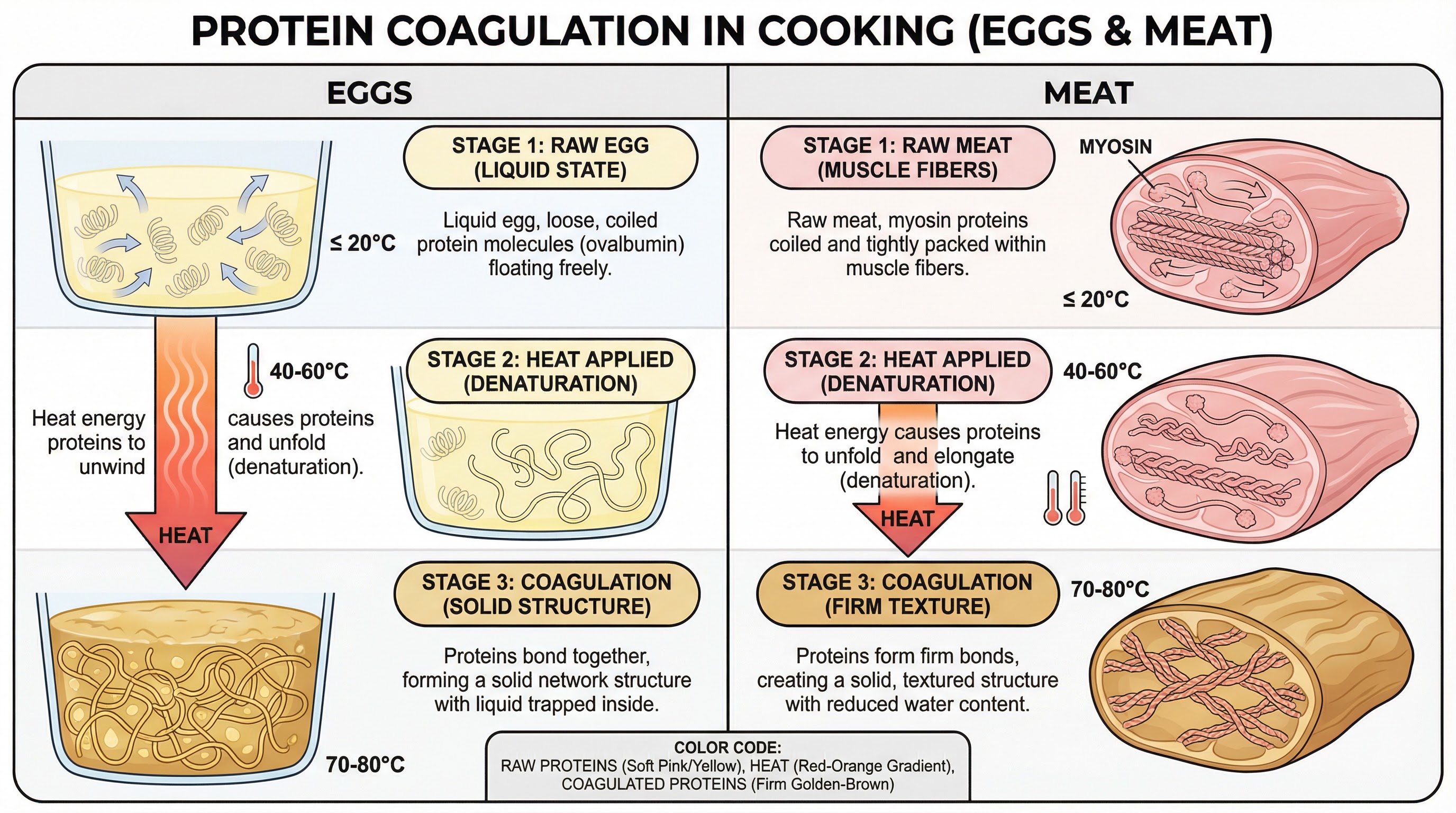
- Process: Proteins are heated or exposed to acid.
- Reaction: The long, coiled protein chains first unwind (denaturation). As heating continues, these unfolded chains bond together, forming a solid 3D network that traps water.
- Outcome: The food changes from a liquid or soft state to a solid, firm texture. For example, a liquid egg becomes a solid fried egg.
Fats: Shortening & Emulsification
Shortening: This is the ability of fat to create a crumbly, flaky texture in pastry and biscuits.
- Process: Fat is rubbed into flour.
- Reaction: The fat coats the flour particles, creating a waterproof barrier. This prevents the flour's proteins (gliadin and glutenin) from absorbing water and forming long gluten strands.
- Outcome: A 'short', crumbly texture rather than a chewy, elastic one.
Emulsification: This is the process of mixing two liquids that do not normally mix, like oil and water.
- Process: An emulsifier (like lecithin in egg yolk) is used to create a stable mixture.
- Reaction: The emulsifier molecule has a hydrophilic (water-loving) head and a hydrophobic (water-hating) tail. The tail embeds in the oil droplets, while the head faces the water, preventing the oil droplets from coalescing.
- Outcome: A stable, smooth mixture, such as mayonnaise or hollandaise sauce."